3D Printed Haversian Bone: Biomimicking for Cell Regeneration
Chinese researchers continue to take on the challenge of bone regeneration in tissue engineering, sharing their findings in the recently released ‘3D printing of Haversian bone—mimicking scaffolds for multicellular delivery in bone regeneration.’
The fabrication of scaffolds is at the center of successful bone regeneration, but this is an area of medicine that is still known to be difficult—despite a wide range of research projects performed already, from using variations in nanotubes to structures with controlled antibiotic release, alternative materials and coatings.
As the authors point out, while bioprinting is an area of medical research where scientists have made huge strides, there are several reasons that obstacles remain:
- Complexity of hierarchical structures
- Mechanical property requirements
- Diversity of bone resident cells
Most regeneration occurs in cancellous bone, however, so this is what researchers must focus on for success in treating patients:
“Cancellous bone is a meshwork consisting of plate-like or rod-like structures at about 200-mm thickness,” explained the authors. “By one estimate, 80 percent of bone remodeling processes occur in cancellous bone. However, bone regeneration not only needs to reconstruct bone structure but also involves repairing other tissues like blood vessels or nerves.”
Biomimetic structures are showing great potential as ‘high-performance bone tissue engineering biomaterials,’ however, leading the research team to focus on the use of 3D printed Haversian bioceramic structures which can imitate bone in a ‘simple but versatile design.’
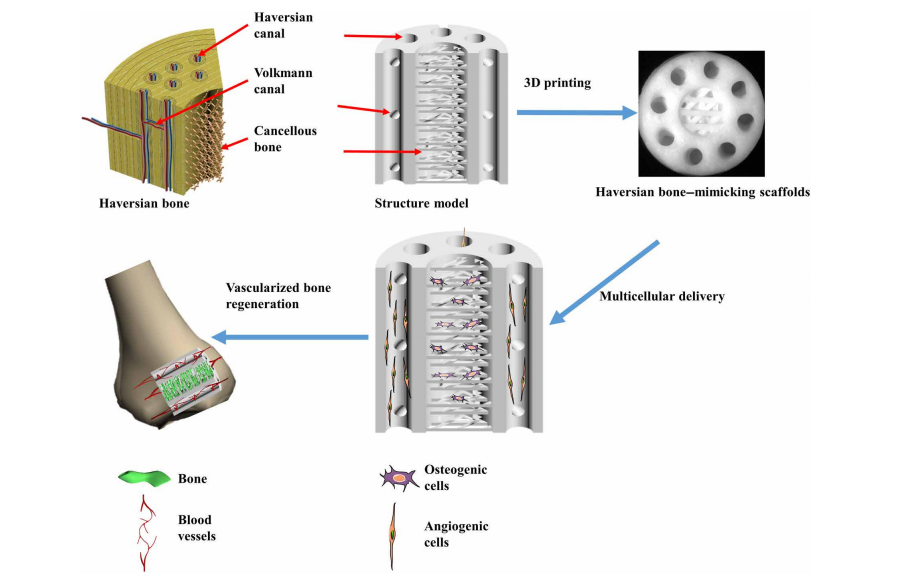
3D printing Haversian bone–mimicking scaffolds integrated with Haversian canals, Volkmann canals, and cancellous bone structure for delivery of osteogenic and angiogenic cells. Osteogenic cells were seeded in cancellous bone structure of scaffolds, and angiogenic cells were seeded on Haversian canals. The Haversian bone–mimicking structure–based multicellular delivery system contributed to the formation of new bone and new blood vessels.
Due to superior osteoconductivity and osteoinductivity, materials composed of akermanite were used in fabrication of five samples with varying measurements in canals and cancellous bone serving as meshwork.
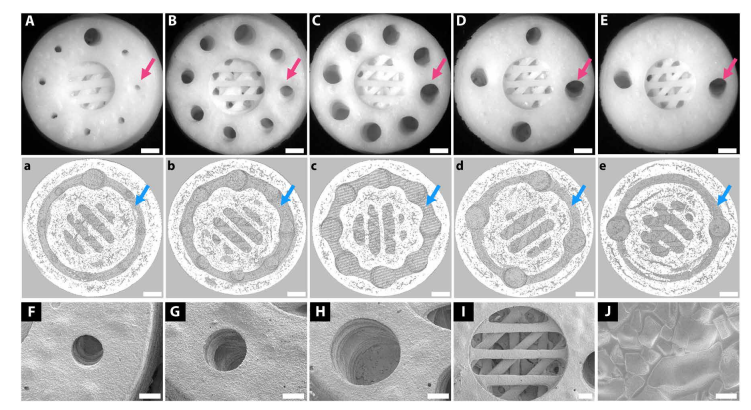
3D printing of Haversian bone–mimicking bioceramic scaffolds with cortical bone and cancellous bone structure. Cortical bone structure contained Haversian canals and Volkmann canals. (A to E) Optical microscope images exhibited different diameters (D) and numbers (N) of Haversian canal indicated by magenta arrows (A) N = 8, D = 0.8 mm; (B) N = 8, D = 1.2 mm; (C) N = 8, D = 1.6 mm; (D) N = 4, D = 1.6 mm; and (E) N = 2, D = 1.6 mm. Scale bars, 1 mm. (a to e) Micro–computed tomography (CT) images show Volkmann canals (blue arrows) connecting Haversian canals in the interior of scaffolds. Scale bars, 1 mm. (F to J) SEM images presented the microstructure of the scaffolds.
“Interconnected Haversian canals were isolated from the cancellous bone structure for noncontact cell coculture,” stated the researchers. “Furthermore, the periphery and bottom of the scaffolds were sealed so that the scaffolds could be used for holding the seeded cells.”
Several different techniques were necessary in creating the scaffolds, to include using electrospinning and twin-screw extrusion, modular tissue engineering, and 3D printing. The team had the most success with DLP 3D printing, designing a series of structures with Haversian canals, Volkmann canals, and cancellous bone.
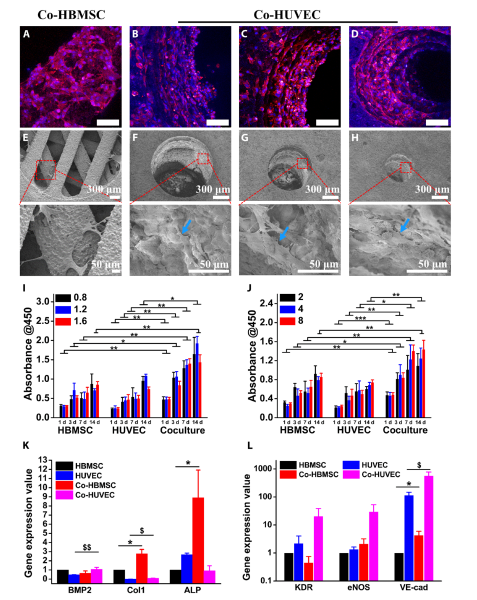
Haversian bone–mimicking bioceramic scaffolds for the HBMSC-HUVEC coculture system performed better in cell proliferation and angiogenic differentiation than monoculture. (A to D) CLSM images of HBMSCs seeded on the cancellous bone structure (A) and HUVECs seeded on the Haversian canal with different diameters, (B) D = 1.6 mm, (C) D = 1.2 mm, and (D) D= 0.8 mm. Scale bars, 100 m. (E to H) SEM images of (E) HBMSCs seeded on the cancellous bone structure and HUVECs seeded on the Haversian canal with different diameters of (F) 1.6 mm, (G) 1.2 mm, and (H) 0.8 mm. (I and J) The proliferation activity of HBMSC, HUVEC, and cocultured HBMSC-HUVEC seeded on scaffolds with different (I) diameters and (J) numbers of Haversian canals after culturing for 1, 3, 7, and 14 days. n = 6 replicates. (K and L) The osteogenic (K) and angiogenic (L) gene expression of HBMSC, HUVEC, Co-HBMSC (HBMSCs in HBMSC-HUVEC coculture), and Co-HUVEC (HUVECs in HBMSC-HUVEC coculture) for 3 days. n = 3 replicates. *P < 0.05, **P < 0.01, ***P < 0.001, $ P < 0.05, $$P < 0.01.

Haversian bone–mimicking bioceramic scaffold–based rBMSC-rSC coculture system performed better in cell proliferation and neurogenic differentiation than monoculture. (A to D) The CLSM images of rBMSC in the (A) rBMSC monoculture group and rBMSC-rSC coculture group with the ratio of rBMSC to rSC being (B) 3:7, (C) 5:5, and (D) 7:3 seeded on the cancellous bone of scaffolds. Scale bars, 50 m. (E to H) The CLSM images of rSCs in (E) the rSC monoculture group and rBMSC-rSC coculture group with the ratio of rBMSCs to rSCs being (F) 3:7, (G) 5:5, and (H) 7:3 seeded on the Haversian canal of scaffolds.
“Here, we further established the Haversian bone–mimicking scaffold–based rBMSC-rSC coculture system with rBMSCs grown in cancellous bone structure and rSCs grown on Haversian canals. Our results indicated that the rBMSC-rSC coculture system exhibited a better proliferation and a higher expression of NGF, BDNF, TrkA, and S100 as compared to rSC monoculture,” concluded the researchers. “It was found that BMSCs could promote SC proliferation and were traditionally used to facilitate the recovery of sensory system.
“Considering the clinical applications of Haversian bone–mimicking scaffolds, there are still some issues to be studied. First, more bone-resident cells such as osteoblasts, osteoclasts, and macrophages should be further considered in the coculture system. The mechanism of multicellular synergistic effects is not fully understood. Further studies are needed to identify the individual effects of coculture cells on the formation of new bone, blood vessels, and nerves in the Haversian bone–mimicking scaffold–based coculture system.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘3D printing of Haversian bone—mimicking scaffolds for multicellular delivery in bone regeneration’]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Astrobotic Tests Rocket Engine Made with Elementum 3D Materials
Astrobotic has completed a series of hot-fire tests for its Chakram rotating detonation rocket engine, with additive manufacturing (AM) playing an important role in how the engine was built. The...
ARC & ORNL Form Partnership to Accelerate AI-Enabled Manufacturing for Defense
Last year, Autonomous Resource Corporation (ARC) became the surprising owner of Desktop Metal’s (DM’s) assets following the bankruptcy of the one-time additive manufacturing (AM) unicorn, an acquisition that cost ARC...
HP Stock Jumps on 3D Printing Buzz
HP (NYSE: HPQ) had its best day in over a year this week, with shares jumping more than 7% on Tuesday. Interestingly, the move was quickly tied to 3D printing,...
HP’s MJF 1200 Targets Entry-Level AM — And Could Shift the Competitive Landscape
HP has launched an entry-level MJF solution, the HP Multi Jet Fusion 1200 3D Printer Solution, which will be available in 2027. What is the 1200 exactly? What does it...










































