For the second year running, and its third year total, 3DPrint.com and SmarTech Analysis have brought the Additive Manufacturing Strategies summit to Boston. With a theme of “The Business of 3D Printing,” the event continues its established coverage of 3D printing in the medical and dental industries, but adds a new metals track this year.
For the second year running, and its third year total, 3DPrint.com and SmarTech Analysis have brought the Additive Manufacturing Strategies summit to Boston. With a theme of “The Business of 3D Printing,” the event continues its established coverage of 3D printing in the medical and dental industries, but adds a new metals track this year.
Lawrence Gasman, the President of SmarTech, welcomed everyone to the event, and then we jumped right into the thick of things, as Dr. Banu Gemici-Ozkan, Senior Market Intelligence Leader for GE Additive, presented her keynote, entitled “Metal Additive Strategies Enabling Next Generation of Adopters.”
Dr. Gemici-Ozkan explained that she’s been working with additive manufacturing for about four years, and her role is to oversee global operations, as well as support business in the metal AM space with the right applications.
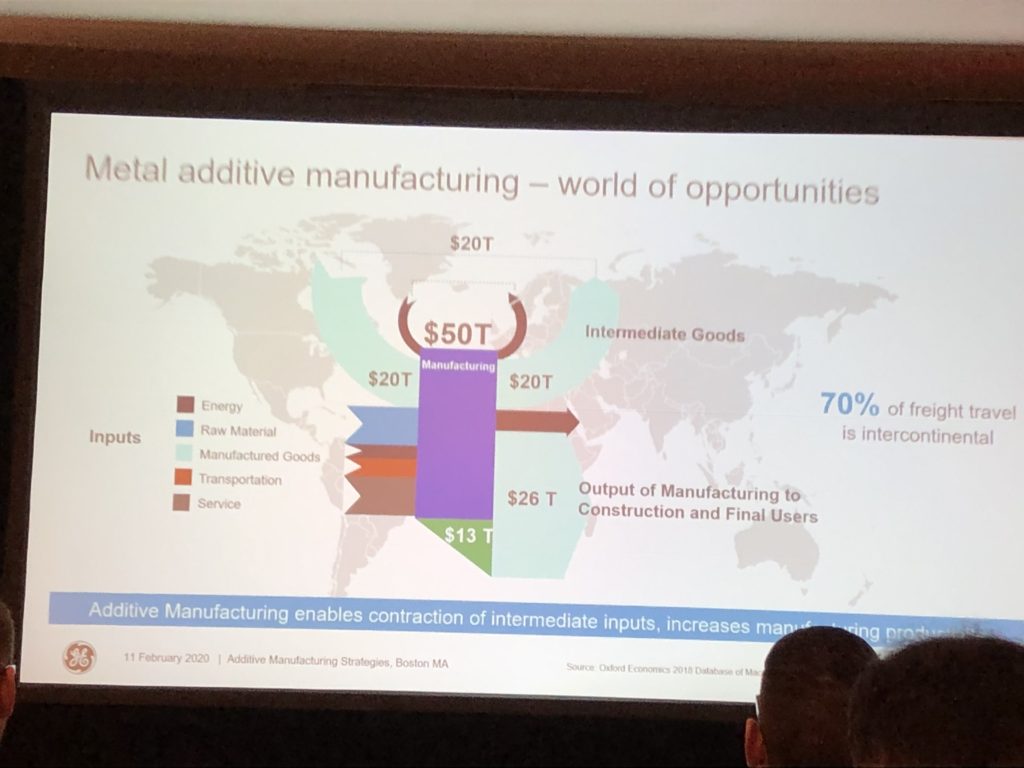
“I’m in marketing, so I have to start with numbers,” she said, pulling up a slide of the “world of opportunities” for metal AM.
She explained that conventional manufacturing happens in many stages – you have to extract the metal, process it in chemical plants, assemble it into the final products, and several others that I’m definitely leaving out. Additive manufacturing can accomplish all of this in less steps, which is why it’s so attractive.
An example of an engine turbine came up, and at the bottom was a statement about how metal AM is competing with $570 billion worth of core conventional metal manufacturing processes. But, system redesign is what makes it competitive to this traditional methods – AM offers a simpler supply chain and leaner operations.
“It’s really exciting to see the potential of additive manufacturing,” Dr. Gemici-Ozkan said. “But where are we in this vision today?”
A timeline showed that the number of metal AM system installations in the first stage of the “diffusion of innovation,” in the 1990s, was less than 50…only the true innovators will put in the work of debugging these first systems and working out the kinks. The early adoption visionaries come in later, excited to invest in the technology.
“The customers are who drive the change,” she said. “So far, we’ve only seen innovators and visionaries.”
She explained that the next generation of the market will consist of the bigger players, or pragmatists, jumping on board. These adopters are cost-conscious, and will be looking for full solutions.
Then, she walked us through what she called the four “critical industries” in metal additive manufacturing. I’m sure you can guess them: medical, dental, aerospace, and automotive. When asked if they were there with the medical field, nearly half the hands in the room were raised, making Dr. Gemici-Ozkan’s point that this sector is a “great space to be in from a metal AM perspective.” The adoption drivers in this industry are cost and performance, with major applications in porous, biocompatible structures with fine features. Here, accuracy, repeatability, and traceability become really important.
Dental is the most mature industry for metal AM, a point that I heard multiple times throughout the day in different presentations. She explained that adoption drivers are lead time and customization; in this and the medical industry, the turnover time with metal 3D printed parts is roughly 24 hours, which you just can’t beat. Additionally, technology providers are focused on meeting customer needs.
In the aerospace industry, industrial production is the main focus. The materials are more versatile, and applications are in large parts and complex geometries with fine features.
“I could talk for hours about this industry,” she said.
“The potential is huge…this space offers a great potential from the industrial production perspective.”
She brought up the GE9X jet engine, which has 304 3D printed components and offers GE Aviation fuel savings of 10% when compared to its predecessor, the GE90, which only featured one 3D printed part.
The automotive industry is already automated, so its needs are focused on cost-conscious systems. Dr. Gemici-Ozkan said that AM technology providers “need to consider integrating their systems to the factory solutions.” The technology will have greater potential in this sector as material costs continue to come down, and she noted that binder jetting will be important in this space.
“Additive manufacturing is not a one-size-fits-all solution – it offers different solutions for different industries and applications,” Dr. Gemici-Ozkan said in summary. “It sounds like it’s all versatile, but these are the building blocks of mainstream technology.”
Then it was time for the next keynote presentation, “Medical 3D Printing: Building the Infrastructure for Innovation,” by Lauralyn McDaniel, Industry Manager, Analysis, for the American Society of Mechanical Engineers (ASME). Part of ASME’s mission is to improve people’s lives through engineering, which is definitely what 3D printing is working towards in the medical field.
McDaniel also started with numbers, with a slide stating that over one million patients had been directly impacted by AM, and that number increases to over two million when you take into account indirect impacts.
“Understanding the history of additive manufacturing in the medical industry can give us clues as to where we go from here,” McDaniel said, before launching into a brief timeline that began with the first 3D printed model from a medical image in 1988.
She explained that some of the factors leading to growth of the technology in the medical field include improved software, more material choices, precision medicine, faster and more precise processes, and the fact that more people share their resources and experience.
“You need published studies to generate the evidence that doctors need,” McDaniel explained.
Challenges include process bottlenecks, verification and validation processes, standards and regulations, and the workforce development.
Then, she cleared up something that many don’t always understand – most materials that people say are FDA-cleared are not, they have just been used in FDA-cleared devices. For example, titanium is often used in orthopedic implants, but the material itself is not cleared by the FDA, it’s just been cleared for use in the implant.
Continuing on to the regulatory process, McDaniel explained that there’s a “big difference” between a new product, and a new way to make the same product.
“The dental industry has a whole infrastructure set up to match patients with devices and implants, 3D printing just gives them a new, more efficient way to do it,” she said. “But anatomical models is a whole new product category.”
McDaniel said that ASME is supporting a series of discussions about the FDA’s concept framework for 3D printing at the point-of-care, and has worked with the agency to create validation and verification standards, including those for 3D printed medical devices. Just over half of the medical devices that have been cleared by the FDA are metal, so never fear, polymers are still significant in this space.
On the clinical side of things, standards aren’t quite as common, but she mentioned that the RSNA Special Interest Group is working to develop guidelines to help others with their own processes.
Some of the development highlights that McDaniel touched on include 3D printing-enabled tissue fabrication, clear dental aligners, which “exploded a bit because some of the patents expired,” tissue fabrication in outer space, and the fact that nearly 150 3D printed medical devices have been cleared by the FDA overall; at least three of these were patient-specific.
Moving forward with medical 3D printing, McDaniel said we need more collaboration and sharing of our experiences and resources, along with continuing materials development, improved software and AI, increased standards development, and more regulatory clarification, especially in hospitals.
Stay tuned to 3DPrint.com as we continue to bring you the news from our third annual AMS Summit.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
[Photos: Sarah Saunders]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Fathom Struggles in Financial Quicksand During Critical Transition
Facing a year of key transitions and financial pressures, Fathom (Nasdaq: FTHM) has filed its annual report for 2023 with the U.S. Securities and Exchange Commission (SEC). The document outlines...
Latest Earnings Overview for Australian 3D Printing Firms Titomic and AML3D
Australian 3D printing manufacturing firms Titomic (ASX: TTT) and AML3D (ASX: AL3) reported their financial results for the period from July to December 2023, marking the first half of their...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...
3D Printing Financials: Unpacking Farsoon and BLT’s 2023 Performance
In the Chinese 3D printing industry, two companies, Farsoon (SHA: 688433) and Bright Laser Technologies, or BLT (SHA: 688333), have recently unveiled their full-year earnings for 2023. Farsoon reported increases...