Influence of High Disperse Active Pharmaceutical Ingredient Loads in Filaments Studied
In the recently published ‘Influence of High, Disperse API Load on Properties along the Fused-Layer Modeling Process Chain of Solid Dosage Forms,’ German researchers delve further into the mechanical properties of materials, 3D printability, and both dosage and release profiles in relation to fused layer modeling with patient-specific treatment.
3D printing in the pharmaceutical realm continues to progress due to speed in production, affordability, and the ability to produce individual doses. Spritam®, produced by the ZipDose® technology, gained FDA approval in 2015, leading to many projections regarding changes to the pharmaceutical industry—with a mixture of excitement, positivity, and concerns too from researchers and 3D printing analysts.
As the researchers point out, there are only several types of doses approved, with one dissolution profile. Selective laser sintering (SLS) 3D printing has also been used for producing doses that disintegrate orally, as well as modified-release tablets. Extrusion-based techniques include selective deposition of hydrogels carrying pharmaceuticals. Called by some FLM (fluid layer modeling) has also been used in pharmaceutical development and is noted as one of the most popular techniques for that type of research.
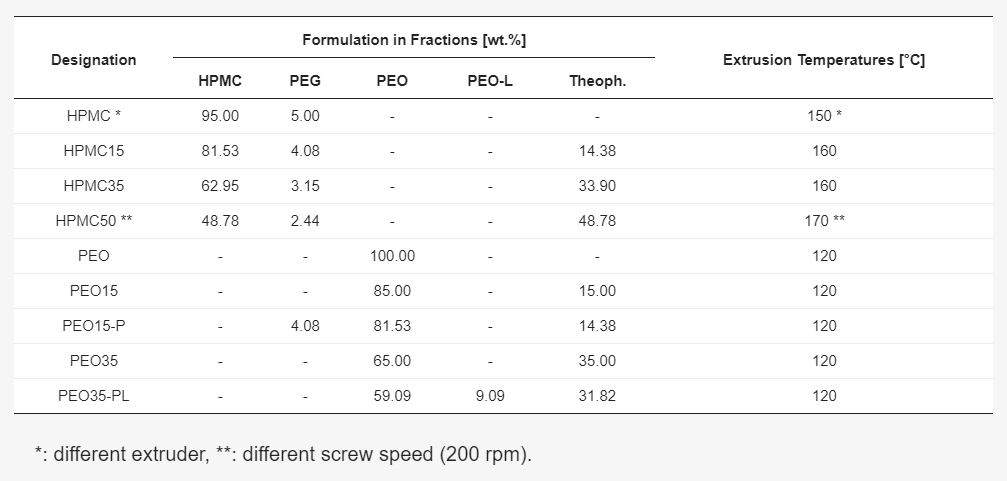
In creating API-loaded filaments, the researchers used different machinery, creating filaments with extruded powder blends, and a single-screw extruder. They experienced ‘marginal shear’ and ‘melt mixing’ and with a DIY ram-extruder, no melt mixing either. For most research, however, they would recommend a co-rotating twin-screw extruder for ‘well-compounded, homogeneous filaments.’
Different API-loaded (API is used for active pharmaceutical ingredient) filaments were used in FLM to create immediate, controlled-release doses, begin with the use of a dual FLM machine. For this study, they introduced theophylline as a model drug at different concentrations. Filaments and forms were then evaluated regarding parameters like:
- Mechanical properties
- Content
- Mass uniformity
- Drug release
Theophylline distribution in filaments was proven with 100 mg samples taken from every 10 cm from the filament, with five at each end of every roll. Samples were then dissolved in 0.1 N HCl and the theophylline concentrations were determined via UV/Vis spectrometry. Research here showed that polymers and additives did not have any effect on measuring, and content uniformity of 3D printed material cylinders was handled in a similar manner. The filament was successfully produced, and ultimately all the raw materials exhibited ‘thermal stability.’ Theophylline was the first material, however, to degrade at above 200 °C.

(A) Images of 3D-printed theophylline-loaded dosage forms with the same volume; (B) SEM image of the surface of a 3D-printed object made from HPMC15 filament.
“In general, particles inside the polymer matrix proved to particularly alter the mechanical properties of both filaments and 3D prints. The systematic adaption of specific FLM parameters can attenuate several challenges such as layer adhesion to a certain extent. However, as the influence of size and shape of particles and their behavior during multiple heating, such as in filament extrusion and FLM itself, and their interplay with dissolved API molecules in the melt are not fully understood, further investigations are required,” concluded the researchers.
“Additionally, higher API content can cause instabilities in the 3D-printing process, making prediction of dosage form mass and therefore content uniformity more challenging. For tested HPMC formulations, the release profile of geometries with low surface-area-to-volume ratio is independent of the API content, facilitating an independent adjustment of the dose-and-release profile as well as the application of highly loaded polymer formulations in polypill applications.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
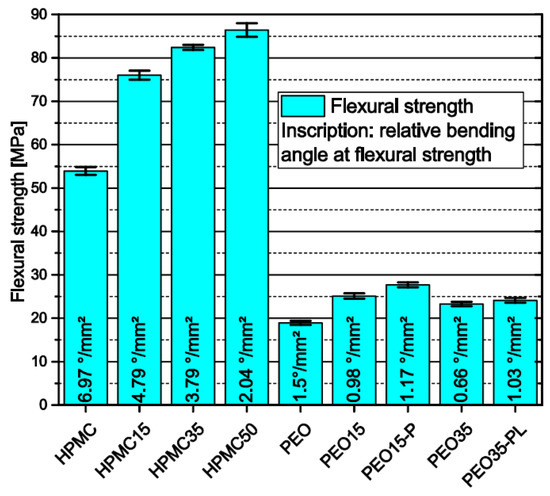
Flexural strength and relative bending angle of filaments measured via three-point-bending span = 16 mm; n = 6.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
You May Also Like
3D Printing Financials: Prodways Ends 2024 with a Profit
After a tough couple of years, Prodways (EPA: PWG) is starting to bounce back. The French 3D printing company finally made a profit in 2024, improved its operating performance, and...
Blue Origin & Auburn University Use EOS M290 to Study Copper 3D Printing
Blue Origin, the commercial space company built off of investments from Amazon founder Jeff Bezos, has donated two EOS M290 powder bed fusion (PBF) printers to Auburn University’s National Center...
Rocket Lab to Acquire Restructured Laser Communications Provider Mynaric AG
Rocket Lab USA, the Long Beach-based, end-to-end space services company that specializes in producing rockets with additive manufacturing (AM), has announced plans to acquire Mynaric AG, a German provider laser...
3D Printing Financials: Stratasys Ends 2024 with Cost Cuts and Growth Plans
Stratasys (Nasdaq: SSYS) has wrapped up 2024 with stronger margins but a full-year net loss. The polymer 3D printing leader navigated a year of economic headwinds, restructuring efforts, and shifting...