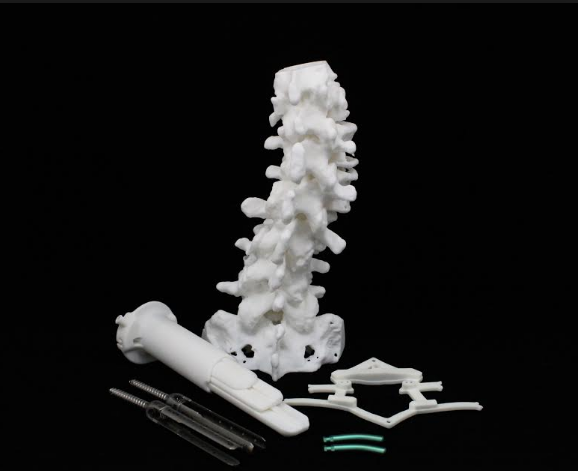
Anatomics Tests 3D Printed SpineBox with 129 Patients
Australia’s Anatomics has been a leader in 3D printing for healthcare long before such technology hit the mainstream, garnering worldwide public attention. Founder Dr. Paul D’urso, a neurosurgeon at Epworth Healthcare, has been involved in 3D printing research since the 90″s and began putting digital fabrication to use as far back as 1995.
Recently, 129 patients have benefited from use of the 3D printed SpineBox. Created to set new standards for both efficiency and quality in spinal fusion procedures, the 3D printed kit is behind the simplification of lumbar fusion surgery.
“SpineBox is a disruptive technology that facilitates communication, planning, education and informed consent to deliver efficiency and quality in spinal surgery” said Paul D’Urso. “By facilitating planning and surgery, SpineBox eliminates the need for complex equipment in the operating room.
“By embedding metrics in the process SpineBox allows validation and verification of efficiency and quality.”
The Anatomics team integrates CT data with their custom planning software, resulting in a comprehensive BioModel of the patient’s spine, along with a muscle retractor, and an anatomically matched SpineGuide template. Titanium plates are required for patient care and are delivered to the medical facility before any procedure occurs.
In a recent press release, the Anatomics team explains that these 3D printed medical instruments facilitate the following:
- Nerve decompression
- Fusion cage implantation
- Accurate completion of the spinal construct
“The pre-planned specifications for the spinal construct can be mapped into the bony spine from the skin surface using the SpineGuide template and Kirschner wires, with minimal radiography. Once deployed, the Kirschner wires provide a railroad for all subsequent instrumentation. The SpineTube muscle retractor is manufactured to match the skin to spine depth is then temporarily affixed to the spine with an insert and locking screw. The unique insert doubles as an osteotomy guide,” explains the Anatomics press release.
A paper authored by Ganesha K. Thayaparan, Mark G. Owbridge, Matthew Linden, Robert G. Thompson, Philip M. Lewis, and Paul S. D’Urso has recently been published on the subject too. In ‘Measuring the performance of patient-specific solutions for minimally invasive transforaminal lumbar interbody fusion surgery,’ we are able to learn more about the value of pre-surgical planning with 3D printed models. In their experience with 129 consecutive patients, the Anatomics team was able to study treatment over 27 months, in one facility, with one surgeon performing the procedures.
According to the research, efficiency measures included:
- Operating time
- Sterile tray usage
- Fluoroscopy screening time
- Operative waste
- Median hospital stay
Accuracy of planning was evaluated by considering proper numbers of screws and rods used, whether interbody case measurements matched intraoperative data, implant size discrepancies, screw placements, and overall deviations.
Results upon viewing data from postoperative CT scans showed that pedicle screw placement was accurate 97.8 percent of the time, although there was one intraoperative dural tear, and six reoperations were required.
“No patients required conversion to midline, mini-open, or open techniques for pedicle screw placement, neural decompression or interbody work. No patients required computer-assisted imagebased navigation, robotic assistants, 3D fluoroscopy, or intraoperative computed tomography to supplement SpineBox equipment in this series,” stated the researchers in their paper
“All patients were mobilized day one post-operation. Two patients had urinary catheters placed post-operatively for urinary retention. The median hospital stay was four days. Four patients were readmitted to revise implant positions. Two patients underwent wound washouts for hydroxyapatite granulomas at the iliac donor site from the use of an injectable hydroxyapatite bone substitute. Subsequent patients received a tricalcium phosphate putty for their iliac donor site without complication.”
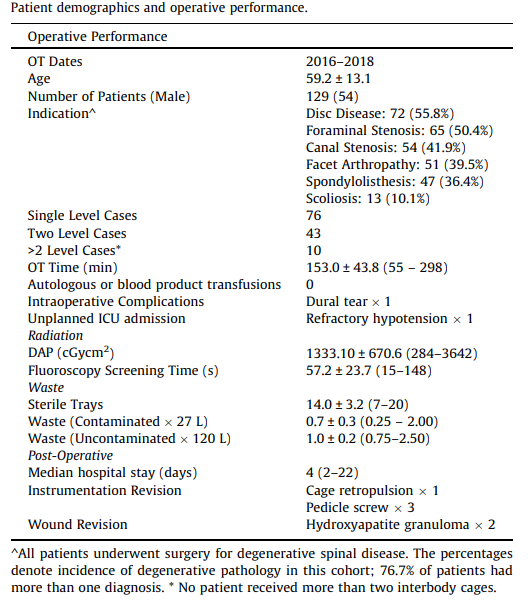
Patient demographics and operative performance. (Image: ‘Measuring the performance of patient-specific solutions for minimally invasive transforaminal lumbar interbody fusion surgery’)
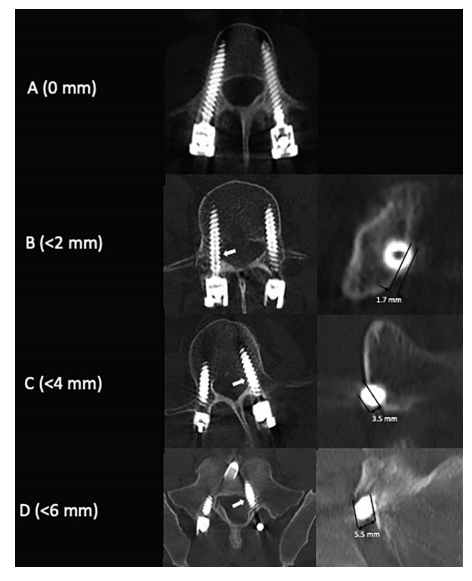
CT scans demonstrating the Gertzbein and Robbins classification system for pedicle screw placement. The grading system measures the deviation of the pedicle screw from the ideal intrapedicular trajectory. Grade A pedicle screws do not breach the cortical bone of the pedicle; Grade B pedicle screws breach the cortical bone by <2 mm. Grade C and D pedicle screws breach by <4 mm and 6 mm, respectively. Grade E (not depicted) pedicle screws breach the cortical bone by >6 mm at any point along its intrapedicular course. (Photo: ‘Measuring the performance of patient-specific solutions for minimally invasive transforaminal lumbar interbody fusion surgery’)
The kits are made available two weeks before surgery. It takes the Anatomics team three days to design the tools, verify, sterilize, and package them. They are helpful not only in treatment but also in education for patients and their families, allowing for a comprehensive device that shows the anatomy of the patient and gives the surgeon a chance to explain in detail with the course of action—especially in surgery—will be. The kits offer true patient-specific solutions and could mean ‘significant cost savings’ in healthcare as the equipment is customized for both the patient and doctor.
“Our experience demonstrates a viable application of patient-specific 3D-printed solutions and provides a benchmark for studies of efficiency in spinal fusion surgery,” stated the researchers.
We have followed Anatomics over the years as they have continued to make strides for this patients, from 3D printing titanium implants to the initial development of their spinal applications for biomodeling, and ongoing support of 3D printing in patient-specific healthcare.

1:1 scale BioModel. (Photo: ‘Measuring the performance of patient-specific solutions for minimally invasive transforaminal lumbar interbody fusion surgery’)
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
Source / Images: Anatomics; ‘Measuring the performance of patient-specific solutions for minimally invasive transforaminal lumbar interbody fusion surgery’]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Colibrium Additive Gets $31 Million NAVAIR Contract
Colibrium Additive has been awarded a $31 million contract by the Naval Air Systems Command (NAVAIR). The contract is part of the Additive Manufacturing Capability initiative, focused on qualification and...
Nikon AM Synergy Secures DLA Contract for On-Demand Military Parts
Nikon AM Synergy has gotten a DLA contract under the JAMA IV IDIQ Pilot Parts Program. The contract with the Defense Logistics Agency (DLA) is a step forward for Nikon...
ExOne Cuts Costs for U.S. Customers as Printhead Production Moves to Detroit
ExOne Global Holdings, created through the 2025 integration of ExOne and voxeljet, is making changes across its U.S. operations. These include starting printhead manufacturing in the Detroit area and lowering...
3D Printing News Briefs, April 8, 2026: LiDAR Scanning, Vapor Smoothing, FDM Optimization, & More
We’ll kick off today’s 3D Printing News Briefs with some 3D scanning news from Artec 3D, and then move on to new America Makes Project Calls. Then, Raise3D and AMT...