A friend tipped me about Owen Hildreth’s work. The Colorado School of Mines Professor has spent years developing an automated metal support removal process for metal printing. This is the holy grail of metal 3D printing. Direct Metal Laser Sintering (PBF) have a huge problem which is that they need support structures to manage thermal stress in the part. These supports are made of metal so have to be cut off by hand. Then the evidence of these supports often has to be filed down manually. This is a significant cost component of metal 3D printing, around two thirds to a third. Manual support removal also introduces the human element into an automated process increasing possible variability and errors. Automated support removal will enable a huge amount of new business cases to emerge for 3D printing. Ping pong ball parts could become softball-sized parts opening up a lot of things people have not yet considered. Parts which now will only work for niche applications may yet be suitable for much larger serial production applications. Removing supports automatically is the holy grail of metal printing and Dr. Hildreth may have solved it.
Dr. Hildreth has demonstrated this in two methods to automatically remove supports with 17-4 PH and 316 stainless steel 3D printed parts:

a direct dissolution approach is demonstrated with the supports electrochemically dissolved. While this process works, it is not self-terminating and maintaining dimensional accuracy for complex geometries is thus difficult. The second approach incorporates a sensitizing agent during the normal stress, relieving thermal annealing step to decrease the chemical stability of the top 100–200 lm of the component surface. The component is then etched under etching conditions with a high selectivity toward the ‘‘sensitized’’ surface over the base component material. This creates an etching process that self-terminates once the sensitized layer is removed.
Direct dissolution was conducted under anodic bias in a solution of HNO3/KCl/HCl; 120 lm of material were removed from the component’s surface. For the self-terminating dissolution process,surface sensitization was done by exposing the sample to sodium hexacyanoferrate at 800!C for 6 h to carburize the top100–200 lm of the sample. This carburization process captures the protective chromium in chromium carbide precipitates and renders the surface sensitive to chemical and electrochemical dissolution. The self-terminating etching reaction was demonstrated under anodic bias in a solution of HNO3/KCl.
120lm of material were removed from the sensitized surface of the component. To further test the self-terminating sensitized approach, a set of 316 stainless steel interlocking rings were fabricated to demonstrate that this approach scales to large components with complex geometry. For these parts, this approach replaced 4–5 days of machining operations with a 32.5 h electrochemical bath.

We interviewed Dr. Hildreth to find out more.
In metal printing, 60% of part cost could be due to support removal and post finishing. Your research could, therefore, have a significant economic impact for us?
We reduce post-processing costs associated with support removal by around 90% to 95%. And can reduce surface roughness down to Ra ≤ 0.4 µm, so surface roughness post-processing costs can also be reduced significantly for surfaces where Ra ≤ 0.4 µm is sufficient.
How does it work?
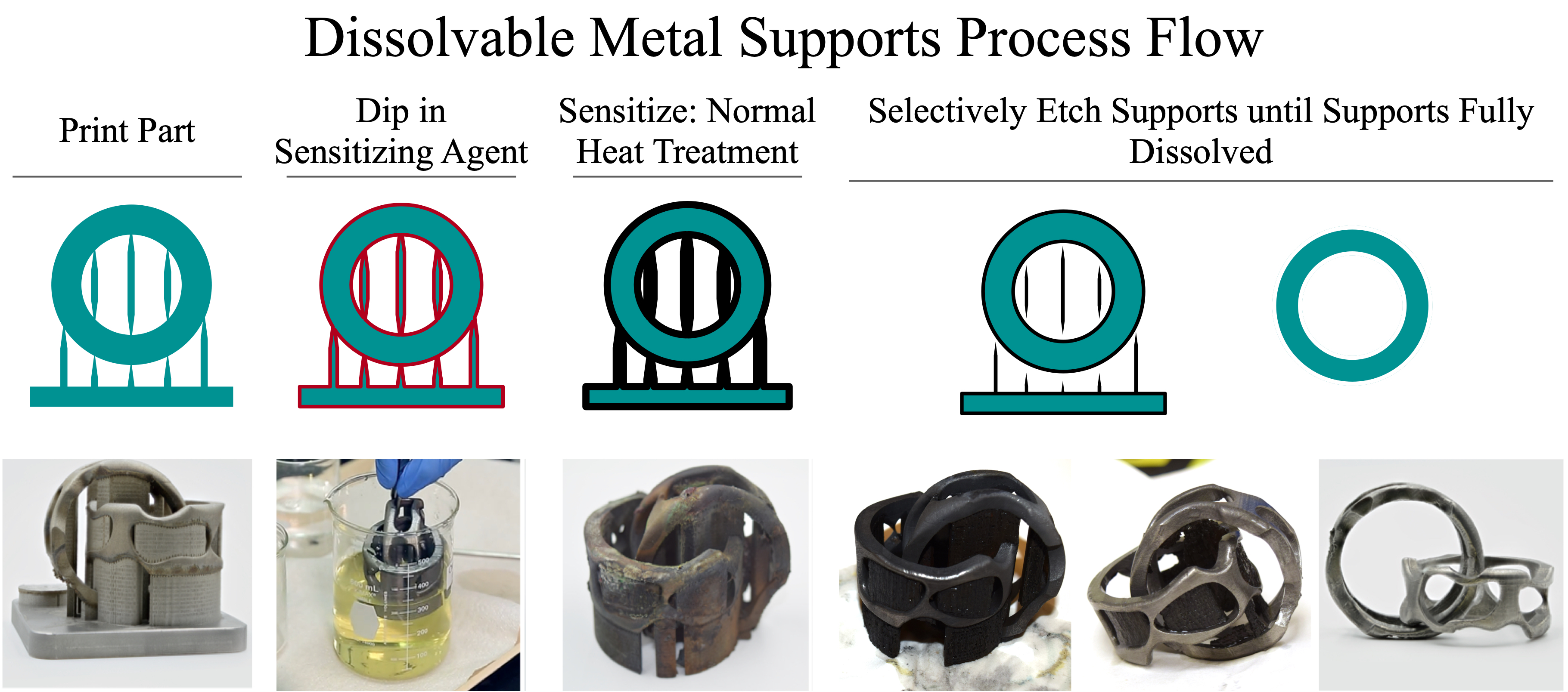
The Process Flow is as follows:
Step 1) Print your part using Powder Bed Fusion (PBF) techniques using existing printers, existing materials, and existing printing parameters. There is NO need to change your existing printing process.
Step 2) Dip your printed part with attached supports and trapped powder into a solution containing a “sensitizing agent.” This coats all of the surfaces with the sensitizing agent (red coating on the image). The sensitizing agents are non-toxic and water soluble.
Step 3) Put the coated part w/ supports into a furnace and run through your normal post-processing heat treatment used to reduce residual stresses and and refine the microstructure.
This is is when the “magic” happens. The high temperatures used during this heat treatment step cause the sensitizing agent to decompose and then diffuse into the top ~100 µm of all the coated surfaces and “sensitizes” the surface.
Effectively, diffusing the sensitizing agent into the surface changes the composition of just that top 100 µm of material. Anytime you have a difference in composition, you have a difference in corrosion sensitivity. Depending on the material system (stainless steel, aluminum, titanium, etc.) we select the sensitizing agent so that it dramatically increases the corrosion sensitivity of the sensitized region. Essentially, we make the top 100 µm of the surface extremely easy to dissolve while the underlying base component material remains extremely corrosion resistant.
Since the supports are pretty thin, < 200 µm in most cases, they are completely sensitized. The same thing with all of the trapped powder and the top 100 µm of the part.
This sensitization process is completely geometry agnostic. All surfaces that were exposed to the sensitizing agent are now, uniformly, sensitized 100 µm
Step 4) Etch away the supports.
We etch the part under conditions where the sensitized region can be etched by the underlying part material can’t. Since the supports where completely sensitized, they are completely removed. The part only looses 100 µm off of its surfaces.
The net result is a simple process that removes all supports and trapped powder while also smoothening all surfaces. This process is simple, requires no change to your existing printer process or settings, and integrates seamlessly existing post-processing heat treatments.
Is it similar to Hirtisation?
I’m not familiar with this process but it looks more like electropolishing to me. As far as I’m aware of, my process is the only one that is geometry agnostic, self-terminating for all surfaces, and doesn’t have any aspect ratio limitations.
So can we do this for all metals or only some?
We’ve published for stainless steel and inconel. We are wrapping up our publications for titanium right now. We will have recipes for Al and GRCop soon too.
It seemed to work much better with carbon steel, are there other materials that are ideal candidates for this? TiAL? Au?
We’ve identified recipes for almost every alloys people have requested. I haven’t thought about Au though.
Could it also possibly work in EBM with gradient materials?
Yes.
With DMLS could we print one part in carbon steel or similar on one machine and then put it in another machine to print a stainless steel on top of it?
Yes. However, that is a different approach compared to the PBF process detailed above.
Could we make a certain type of build plates that would enable easy removal from the build plate? This itself would save labor.
Yes.
What will it take for this to be commercialized?
I have started a company, InnovAMPP to commercialize the process. We are collecting fatigue and corrosion data right now.
How far away is this technology from being available?
6-9 months.
Are you looking for partners?
Yes. We are currently getting investors and have over 30 companies lined up to test out and use our process.
Sometimes parts spend a week or more in a tumbler to finish. Can this be reduced as well? What kind of Ra’s can you get?
We get Ra ≤ 0.4 µm with our Titanium process with 24 hours of etching.
Can you electrochemically etch in a controlled way to get certain surface finishes? As in an etch-predeform? Can you compensate the part size to do this in a controlled way?
Yes, because we only dissolve the sensitized region, we have a self-terminating etching process that is geometry agnostic. We remove the same amount of material on all surfaces, so it is very easy to compensate for the 100 µm we remove.
What other areas of research interest you?
I do a lot of research printing reactive inks instead of particle-based inks and powders. The long-term goal is to be able to print your phone from an inkjet-style printer. We currently have funding to develop reactive silver inks to metallize solar cells and reduce the amount of silver needed to print the top-side contacts by 90%. Currently, silver accounts for ~25% of the materials cost of a solar cell. It should save the photovoltaics industry ~$2 billion/year.
What is holding 3D printing back?
3D printing is really, really close. Companies are investing billions of dollars in the technology and it is finally coming close to be commercially viable for real-world production. Some of the main issues I hear about involve post-processing (which I’m trying to eliminate) and being able to certify printed parts.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Unpeeled: New Arkema Material for HP, Saddle and Macro MEMS
A new Arkema material for MJF is said to reduce costs per part by up to 25% and have an 85% reusability ratio. HP 3D HR PA 12 S has been...
3D Printing News Briefs, January 20, 2024: FDM, LPBF, Underwater 3D Printer, Racing, & More
We’re starting off with a process certification in today’s 3D Printing News Briefs, and then moving on to research about solute trapping, laser powder bed fusion, and then moving on...
3D Printing Webinar and Event Roundup: December 3, 2023
We’ve got plenty of events and webinars coming up for you this week! Quickparts is having a Manufacturing Roadshow, America Makes is holding a Member Town Hall, Stratafest makes two...
Formnext 2023 Day Three: Slam Dunk
I’m high—high on trade show. I’ve met numerous new faces and reconnected with old friends, creating an absolutely wonderful atmosphere. The excitement is palpable over several emerging developments. The high...































