Lawrence Berkeley National Laboratory: Researchers Use Liquid-in-Liquid Printing to Create 3D Fluidic Devices
 The mixing of oil and water is generally something most of us have no use for, with the two known to be immiscible liquids—meaning they do not combine together and eventually separate into layers. Researchers at Lawrence Berkeley National Laboratory are currently studying how these types of mixtures could be helpful in a variety of different scientific applications though. Their findings are discussed in a recently published paper, ‘Harnessing liquid-in-liquid printing and micropatterned substrates to fabricate 3-dimensional all-liquid fluidic devices,’ authored by Wenqian Feng, Yu Chai, Joe Forth, Paul D. Ashby, Thomas P. Russell, and Brett A. Helms.
The mixing of oil and water is generally something most of us have no use for, with the two known to be immiscible liquids—meaning they do not combine together and eventually separate into layers. Researchers at Lawrence Berkeley National Laboratory are currently studying how these types of mixtures could be helpful in a variety of different scientific applications though. Their findings are discussed in a recently published paper, ‘Harnessing liquid-in-liquid printing and micropatterned substrates to fabricate 3-dimensional all-liquid fluidic devices,’ authored by Wenqian Feng, Yu Chai, Joe Forth, Paul D. Ashby, Thomas P. Russell, and Brett A. Helms.
In harnessing liquid structures to create 3D fluidic devices, the researchers explained that such an exercise is an ‘emerging design paradigm’ for chemists today interested in manipulating soft matter and figuring out ways to produce them on demand.
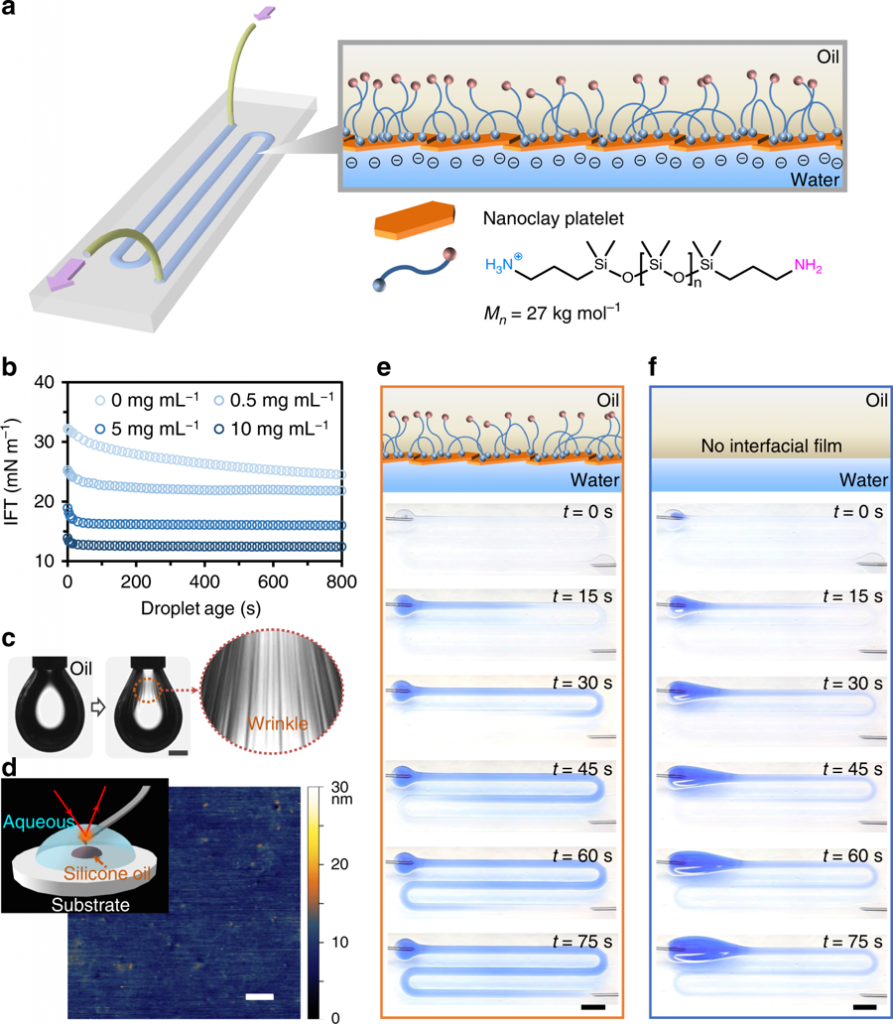
All-liquid fluidic devices stabilized by NPS membranes. a Schematic of an all-liquid fluidic device comprised of immiscible liquid phases confined in space using superhydrophobic−superhydrophilic micropatterned substrates. Nanoclay–polymer surfactants (NPSs) self-assemble at the liquid–liquid interface, forming an elastic wall that allows the all-liquid architecture to maintain integrity while fluid is pumped through the channel. b Temporal evolution of interfacial tension (IFT) of aqueous nanoclay dispersions (0.5, 5, and 10 mg mL−1, pH 7.0) introduced to solutions of H2N-PDMS-NH2 in toluene (10% w/w), illustrating control over the rate of NPS assembly at the interface. c Buckling behavior observed when retracting a droplet cladded with the interfacial NPS film. Scale bar, 1 mm. d In-situ AFM image of the NPS film. The inset shows the schematic diagram of the experimental setup for AFM measurements. Scale bar, 100 nm. e, f Time-lapse images of a solution of blue-colored dye being pumped (10 mL h−1) through the channel in the presence (e) and in the absence (f) of the NPS film. Scale bars, 5 mm. AFM atomic force microscopy
“Still nascent in their development, structured liquids presently lack clear design rules for controlling their 2D or 3D architectures, spatially directing functional components within each liquid phase, and coupling physiochemical processes across the liquid−liquid interface so as to create autonomous chemical systems capable of performing useful work, processing information, or executing logical functions,” state the scientists.
To make such devices, glass supports were coated with superhydrophobic polymers. Next, the researchers used photo-patterning with superhydrophilic channel architectures, also accompanied by an aqueous dispersion of anionic 2D nanoclays.
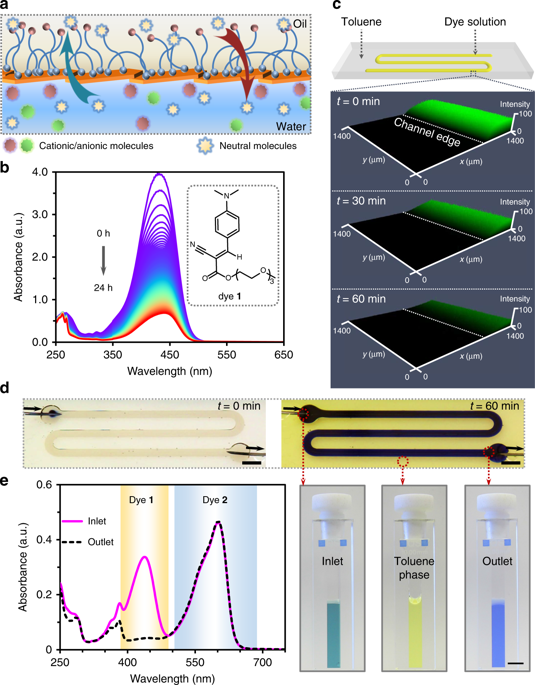
Semi-permeability of 2D NPS interfacial films. a Schematic showing selective mass transfer across the water–oil interface through the NPS film. b Stacked UV–Vis spectra of a neutral dye (1) at 5-min time-intervals, monitoring dye transfer from water to toluene across the NPS membrane film in a masked cuvette. The inset shows the chemical structure of dye 1 engaged in mass transfer. c LSCM images of the fluidic channel after infusing dye 1 solution in the preformed liquid channel covered by toluene. d A fluidic device depicting the flow of a mixed solution of dye 1 and resazurin sodium (2) for chemical separation. [1] = 1.4 mg mL−1, [2] = 1 mg mL−1. Photographs of the diluted inlet, outlet, and overlay of toluene solutions collected in cuvettes show the dye separation after infusing the mixed solution at a flow rate of 0.2 mL h−1 for 1 h. Scale bar, 5 mm. e UV–Vis spectra of the diluted inlet and outlet solutions used to quantify the efficiency of dye 1 partitioning from the aqueous phase to the toluene phase. NPS nanoparticle−polymer surfactant, LSCM laser scanning confocal microscopy
“Interfacial forces are effective in pinning and confining the aqueous phase in arbitrarily complex geometries and a wide range of channel widths,” stated the research team.
The manipulated systems are able to reach the desired steady state quickly, and nanoparticle−polymer surfactants (NPS) topography was presented after the researchers employed atomic force microscopy. They reported that their data demonstrated ‘well-packed nanoclays’ at the interface with no structural problems. The researchers also noted that only microchannels with NPS walls could guide flow at the desired rate. Without them, the aqueous phase proceeded to build up at the channel entrance. The team also noted that maximum flow is completely reliant on the channel’s cross-section and overall architecture.
The team also went on to investigate membrane permeability, functionalization, and further chemical transformations regarding the fluidic devices with the requisite NPS walls. With the use of NPS films, they found that added microchannels were ‘straightforward to introduce’ direct-write methods.
“To contextualize this advance, without the aid of micropatterned substrates and NPS films, constructing channel-like aqueous threads in oil is not typically possible, as the thermodynamic driving force required to reduce interfacial area breaks up aqueous threads into droplets,” stated the researchers.
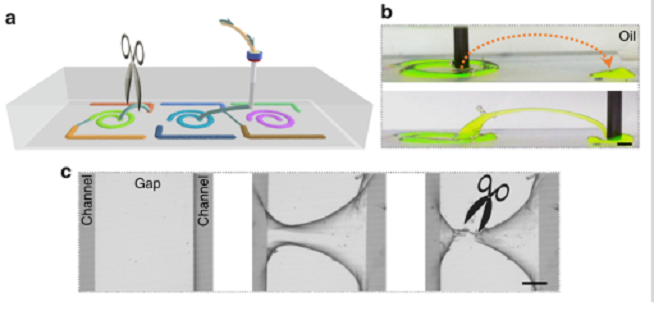
a Schematic of fabricating 3D microchannels stabilized by nanoclay-surfactant interfacial assembly to connect the separated microchannels by printing of water in oil. Disconnecting these microchannels is also possible by cutting these “bridges” off. b Adjacent channels on a patterned substrate connected by a 3D printed overpass. Na-fluorescein (0.1 mg mL−1) has been added to the clay dispersion to clarify features of the constructs. Scale bar: 2 mm. c Fabrication of a 3D bridge linking separate channels (middle panel) and clipping the bridge (right panel). Scale bar: 500 µm.
The fabricated microchannels offered such a stable structure during research and evaluation that the researchers found they could use them as bridges either connecting separate regions on the substrate, or in connecting the device to an exogeneous entity.
“Our studies uncover a latent learning ability in such devices, in that physiochemical sensing or detection of channel properties and contents can be used to direct the architecture of the device to achieve a specific outcome. Maturation of the design concept led to devices that can execute complex tasks in a logical manner by reversible compartmentalization of function and direction of chemo-energetic flows that operate far from equilibrium conditions,” concluded the researchers.
“The potential for this system to exhibit autonomous learning is evident. Such devices may also be arrayed to generate deep or dark data for machine learning, e.g., from all-liquid (bio)chemical transformations and screens, to build knowledge and understanding from chemical logic.”
Fabrications methods used in 3D design and 3D printing today span many different industries and intricate applications, but you may be surprised to explore further into the science of materials, hardware, and software, and realize how many chemists are using the technology from use in continuous flow systems to studies centered around miniaturization and complex designs in microfluidics. Learn more about current processes in liquid-in-liquid printing here.
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: Harnessing liquid-in-liquid printing and micropatterned substrates to fabricate 3-dimensional all-liquid fluidic devices]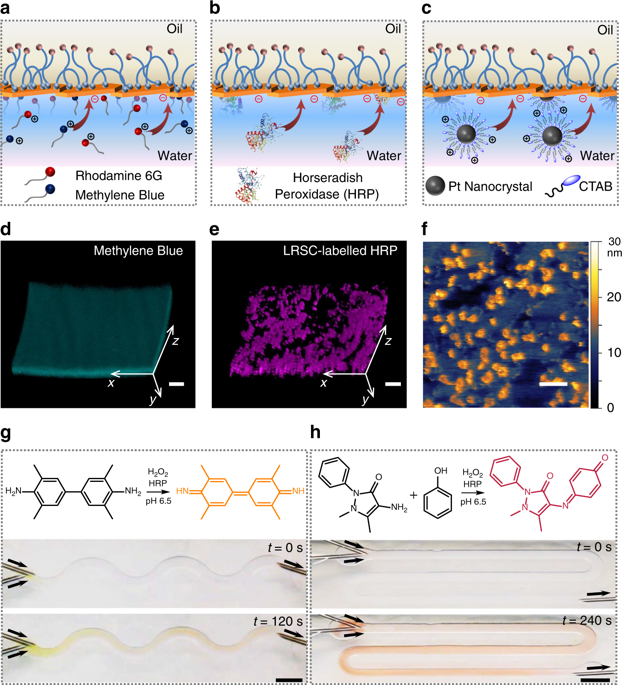
All-liquid microreactors. a–c Schematic of the anchoring of cationic molecules (a), enzymes (b), and nanocrystals (c) to the anionic NPS film lining the microchannels. d, e 3D reconstitution of confocal images of methylene blue (d) and LRSC-labeled HRP (e) to determine the adhesion of cationic species to the NPS wall of the microchannel. Scale bars, 100 µm. f In-situ AFM image of the NPS film immobilized with CTAB-coated Pt nanocrystals at the water−silicone oil interface. Scale bar, 100 nm. g, h Oxidation of TMB (g) and 4-AAP/phenol (h) substrates catalyzed by immobilized HRP in the channels, which generate products with distinctive colors under flow. Scale bars, 5 mm. NPS nanoparticle−polymer surfactant, LRSC lissamine rhodamine B sulfonyl chloride, HRP Horseradish peroxidase, TMB 3,3′,5,5′-tetramethylbenzidine, 4-AAP 4-aminoantipyri
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Flashforge Bets on Meshy AI as Desktop 3D Printing Battle Intensifies
Competition in desktop 3D printing is brutal. Whereas before, firms competed through value engineering, Prusa clones now have an integrated hardware, sensor, and software setup that is making all the...
Ford Uses Binder Jet 3D Printing to Make Boat Propellers for Sharrow Marine
Ford’s Advanced Industrial Technology and Platforms (ATP) group has helped Sharrow Marine make a boat propeller in two weeks rather than 130 days. Thanks to the Michigan Central program, Ford...
Skuld to Work on DARPA’s Rubble to Rockets (R2R) Program
Skuld will work on the Defense Advanced Research Projects Agency’s (DARPA) Rubble to Rockets (R2R) Program, which turns scrap metal into missile components. Skuld will help with alloy design, characterization, and...
From “Magic” to Metal: How Intrepid Automation Wants to Make 3D Printing Matter at Scale
Ben Wynne still talks about 3D printing the way people do when they’ve felt that “wow” moment up close. Back in the early 2000s, he was working at HP’s advanced...

















































