Germany: IMT Researchers Assess 3D Printing Materials & Processes in Miniaturization & Microfluidics
 Just as 3D printing is allowing us to create so many things never before possible, the technology itself continues to expand beyond its previous limitations. Miniaturization is a good example, as explored recently by German researchers at Institute of Microstructure Technology (IMT) in ‘High‐Performance Materials for 3D Printing in Chemical Synthesis Applications.’
Just as 3D printing is allowing us to create so many things never before possible, the technology itself continues to expand beyond its previous limitations. Miniaturization is a good example, as explored recently by German researchers at Institute of Microstructure Technology (IMT) in ‘High‐Performance Materials for 3D Printing in Chemical Synthesis Applications.’
Previously 3D printing resins have not been evolved enough to handle the chemistry required in fabricating microreactors or lab-on-a-chip devices. In this latest study, the researchers study new developments in materials and technique, focusing on transparent silicate glasses, ceramics, and fluorinated polymers.
Here, miniaturization is discussed regarding the field of microfluidics and the potential for creating intricate but in many cases, microscopic, devices. Historically these types of devices are fabricated with silicon, but as the researchers point out, that is difficult to do with 3D printing. As the use of polymers has progressed, the scientists see promise for microfluidics too.
“Additive manufacturing, the layer‐by‐layer generation of a component, holds the great promise of reducing the poly‐mer structuring process to a single step of fabrication using one machine and only requiring a digital model of the desired structure,” state the scientists.
Fabrication of chips and prototypes is already going strong, but what the researchers would like to see happen is for a stronger connection between microfluidics and chemistry. As they point out, 3D printing has also been used successfully for chemical batches in low-volume production, along with flow-through synthesis reactors used for making pharmaceutical molecules. This is generally achieved through stereolithography (SLA) printing though, which is in incompatible for the scientist’s purposes here as it leads to negative chemical reactions. Because of this, the research teams has realized a need for materials in SLA printing that are uniquely and highly resistant. They also see such ‘fine-tuning’ as an opportunity to control further micro- and nano-structuring of bulk material and create ‘significantly enhanced’ materials
“We have recently shown the fabrication of such a material system with the introduction of Fluoropor, an optically transparent fluoropolymer foam that can be structured using 3D printing techniques for generating surfaces with enhanced wetting properties, such as hydrophobic/superhydrophobic patterns,” state the researchers. “Much effort is made to produce such selectively wetted surfaces also for organic compounds to facilitate synthesis in droplets and the fabrication of chemical arrays.”
Creating new SLA printing materials, according to the research team, means mixing the following:
- Polymerization initiators
- Inhibitors
- Monomeric species
All of these together should allow for 3D printing of microstructures in high resolution. In this study, the researchers focus on fabrication with fused silica glass, which they describe as ‘one of the most chemically and thermally stable materials, ceramics, and the most inert type of polymers, fluoropolymers.’

3D printing of ceramics: a) Cellular tube fabricated by stereolithography printing of alumina nanocomposites and subsequent thermal debinding and sintering. Reproduced with permission.45 Copyright 2014, John Wiley and Sons. b) Cork screw printed using preceramic polymers. Reproduced with permission.47 Copyright 2017, Science. c) Robocasted heterogeneous ceramic catalyst fabricated by printing an Al2O3/Cu slurry and subsequent sintering at 1400 °C. Reproduced with permission.52 Copyright 2016, Elsevier.
Fabrication of transparent silicate glasses was not achieved in 3D printing for over 20 years, although the researchers explain that there were many attempts leading to results with white or nontransparent parts. Ceramics have also been explored and are being used in 3D printing in numerous cases we have followed before—from use in printing replacement parts on naval ships to making jewelry to medical implants. As a class of material for 3D printing, they exhibit both high thermal and chemical resistance.
“Here, preceramic polymers with adjustable rheological properties could in future work become an interesting alternative for direct patterning of high‐resolution components for chemical synthesis applications,” state the researchers.
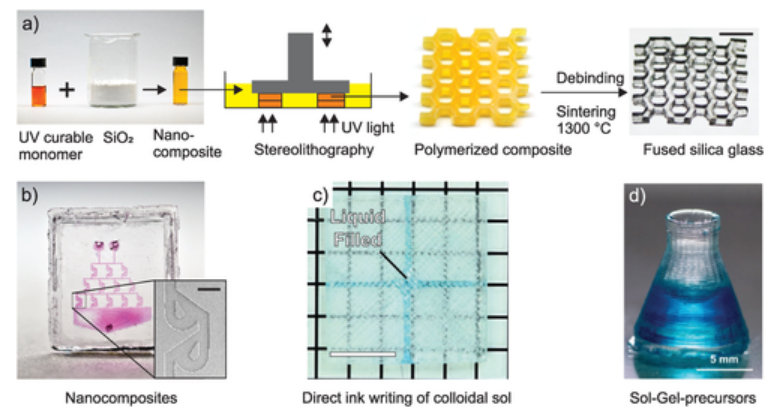
3D printing of transparent glass. a) Stereolithography printing of silica nanocomposites. Amorphous silica nanoparticles are dispersed in an acrylic photocurable binder matrix. The nanocomposites can be printed using stereolithography printers. The printed part is then converted to a transparent fused silica glass via thermal debinding and sintering (scale bar: 7 mm). b) Microfluidic Tesla mixer fabricated using microlithography (scale bar: 200 µm). (a,b) Reproduced with permission.33 Copyright 2017, Springer. c) Cavity filled with dyed water printed using direct ink writing of colloidal silica suspensions (scale bar: 4 mm). Reproduced with permission.36 Copyright 2017, Wiley. d) Erlenmeyer flask printed using hybrid sol‐gel precursors in a stereolithography printer. Reproduced with permission.37 Copyright 2018, American Chemical Society.
In assessing polymers as a rule in 3D printing, the team explains that most of them do not have high chemical stability, but fluoropolymers are different, offering very low surface energy.
“The combination of printed superhydrophobic Fluoropor structures with a hydrophilic surface such as glass allows fabricating patterned surfaces which have gained significant interest in chemical synthesis of rare chemicals in droplets or open surface microfluidic reactors,” state the researchers. “Since the polymerized Fluoropor samples possess a nonstructured top layer due to the polymerization process, superhydrophobic patterns can be fabricated by simply peeling off the last printed structured top layer. Future work will concentrate on superoleophobic/superoleophilic patterns enabling highly parallelized screening platforms for chemical synthesis.”

3D printing fluorinated polymers: a) Teflon part printed using stereolithography and sintering (scale bar: 1 mm). Reproduced with permission.27 Copyright 2018, IEEE. b) Microfluidic mixer structure fabricated by using PFPE methacrylates and commercial stereolithography printers (scale bar: 2 mm). Reproduced with permission.64 Copyright 2017, MDPI. c) High‐resolution woodpile structure in PFPE‐based resin structured using two‐photon polymerization (scale bar: 2 µm). Reproduced with permission.65 2013, American Chemical Society.
The scientists state that 3D printing does offer great potential for on-chip synthesis and includes the usual benefits of speed in production and greater affordability. They conclude by saying:
“Miniaturization also significantly increases experimental throughput due to the inherent reduction in volume, increased thermodynamic control, shorter diffusion distances, and thereby faster reactions. However, most prominent is the potential for massive parallelization which will be key for the exploration and assessment of the chemical reaction space (pressure, temperature, stoichiometry, catalysts, etc.), candidate drug screening as well as optimization of synthesis conditions. To fully leverage these advantages, the design of tailored 3D printing materials will be key.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘High‐Performance Materials for 3D Printing in Chemical Synthesis Applications’]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Air Force Awards Fortius Metals $1.25M to Qualify 3D Printing Wire for Hypersonic Applications
AFWERX, part of the US Air Force Research Laboratory (AFRL), awarded a Direct-to-Phase II Small Business Innovation Research (SBIR) contract worth $1.25 million to Colorado’s Fortius Metals, to accelerate qualification...
US Air Force Awards JuggerBot $4M for Large-format Hybrid 3D Printing
Large-format 3D printer manufacturer JuggerBot has received a $4 million grant to develop a large format 3D printer, courtesy of the Under Secretary of Defense, Research and Engineering Manufacturing Technology...
Where Have All AM’s Unicorns Gone?
In the rapidly evolving world of 3D printing, startups valued at over a billion dollars, known as unicorns, once seemed as fantastical as the mythical creatures themselves. While a few...
How My Childhood Fascination with Planes Led to Investing in 3D Printing
My fascination with aerospace started young, and I started studying planes–identifying them in the sky and learning everything I could about how they work. Fast forward to my first week...































