As 3D printing, additive manufacturing, and bioprinting have offered substantial new avenues for innovation in the medical field and so many other industries, there are bound to be some downsides. And while obstacles in technique often present themselves, hazards regarding safety, emissions, and toxins are often the topic of study. But what about hazards for patients receiving 3D printed implants internally? Researchers Nihal Engin Vrana, Amir Ghaemmaghami, and Pinar Zorlutuna explored this question and more in their editorial ‘Adverse Reactions to Biomaterials: State of the Art in Biomaterial Risk Assessment, Immunomodulation and in vitro Models for Biomaterial Testing,’ while also listing a number of relevant articles on the topic.
A number of adverse reactions can occur when a 3D printed device is implanted into the body, to include:
- Allergies
- Chronic inflammation
- Greater susceptibility to infection
- Collateral tissue damage
- Loss of functionality within immune system
“These concerns have created a general reticence in the medical device industry for the utilization of novel biomaterials and complex, multi-material structures which significantly hinders the advances in the field and also decelerates the introduction of new and potentially transformative technologies to the healthcare system,” state the researchers in their editorial.
The articles they cite as having relevance to this subject overall include topics on skin substitutes, haemocompatibility of biomaterials and the analyses of interactions of biomaterials with human blood, liver- and lung-on-a-chip systems, and an overview of biomaterials.
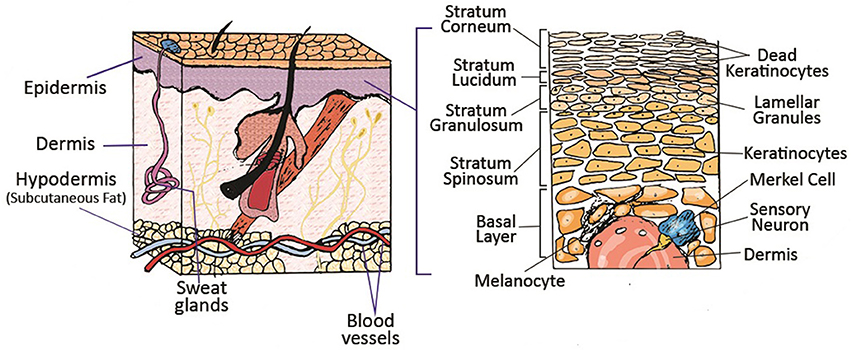
Schematic of different layers of skin and its appendages. Reprinted with permission from Mohammadi et al. (2016). Copyright 2018, John Wiley & Sons.
3D printing has without a doubt had a major impact in healthcare, spanning nearly every aspect of what is undeniably a vast and ever-growing realm. 3D printed medical models are being used more because they have multi-faceted advantages, from helping in diagnosis and treatment to also allowing for educational advances to explain to patients and their families what is happening during an illness or consequent surgery, as well as giving medical students the opportunity to learn about conditions, and train in surgical procedures. Surgeons may also use 3D printed models in the operating room.
Other devices such as 3D printed prostheses have become extremely popular around the world and in developing countries as they can be created so quickly and economically and easily distributed. Bioprinting and biomaterials, however, are a much more serious progression into 3D printing and medicine as the eventual goal, the holy grail for many, is the fabrication of human organs—thus eliminating donor waiting lists, donor rejection in the patient, and a list of other issues that may in some cases mean a shortened life span.
In considering the benefits and the growing list of concerns, the research team suggests improving on risk assessment through in vitro testing, allowing for an internal view of how the human body is interacting with 3D printed implants after they are inserted. They also suggest the use of innovative technology that could act as a controlling mechanism for the 3D printed implant once it is in use within the body.
“The key to better harness the innovations in biomaterial and biomedical device fields is to establish the necessary methodologies and model systems for their risk assessment, validation, and testing,” concluded the researchers. “Recent success and ongoing efforts in developing technologies for immune engineering, personalized biomaterials and personalized in vitro testing platforms will bring forth the solutions that can improve the quality of life and life expectancy even further in twenty-first century.”
Bioprinting and the study of biomaterials continues to expand today, from interests in regenerative medicine and 4D printing to progressive bioinks and materials like microgels. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source: Adverse Reactions to Biomaterials: State of the Art in Biomaterial Risk Assessment, Immunomodulation and in vitro Models for Biomaterial Testing,’]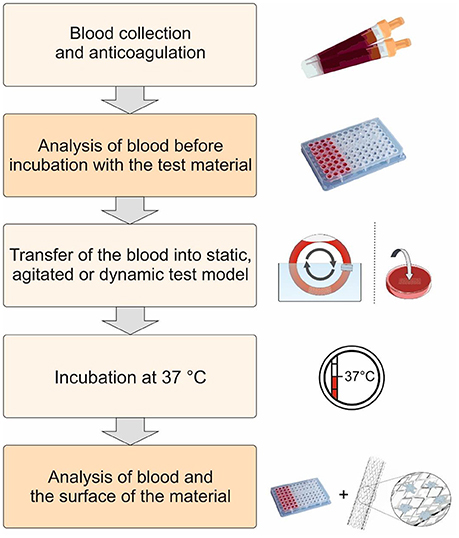
Schematic representation of the procedure for the evaluation of the hemocompatibility of biomaterials. First, fresh human blood is collected and anticoagulated with low dose heparin. Thereafter, the test material is incubated at 37°C using static, agitated, or dynamic test models with the blood. The activation markers in the blood are analyzed before and after the incubation with the test material. Furthermore, the surface of the biomaterial is analyzed to determine the interaction of blood cells and proteins with the biomaterial surface.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...