3D Printed Microfluidic Device Designed to Customize Cancer Treatment
Testing cancer treatments is a lot of trial and error currently, and patients are often subject to multiple uncomfortable and time-consuming therapies before finding one that works. Developments have been made, including growing artificial tumors to test drugs on specific cancer types, but these tumors can take weeks to grow and they don’t account for patients’ individual biological makeup. Now, however, researchers from MIT and Draper University have come up with a new option: a 3D printed microfluidic device that simulates cancer treatments on biopsied cancerous tissue.
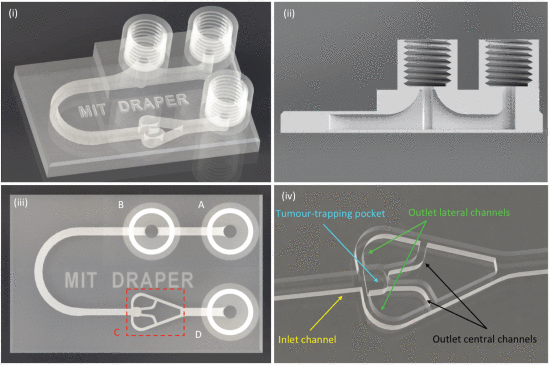
The device is a chip slightly larger than a quarter that can be 3D printed in about an hour. It has three cylindrical chimneys protruding from the surface, which are ports that input and drain fluids as well as remove unwanted air bubbles. The biopsied tumor fragments are placed in a chamber connected to a network of deliver fluids to the tissue. These fluids could contain things like immunotherapy agents or immune cells. Clinicians can then use imaging techniques to see how the tissue responds to the treatments.
The researchers used a new type of biocompatible resin, traditionally used for dental applications, that can support the long-term survival of biopsied tissue. This contrasts with other 3D printed microfluidic drug testing devices, which have chemicals in the resin that quickly kills the cells. Fluorescence microscopy images showed that the new device, called a tumor analysis platform or TAP, kept more than 90 percent of the tissue alive for at least 72 hours and potentially much longer.
The TAP is cheap and easy to fabricate, so it could quickly be implemented into clinical settings, according to the researchers. The devices is adaptable as well – doctors could 3D print a multiplexed device that could support multiple tumor samples in parallel, so that the interactions between tumor fragments and several different drugs could be modeled simultaneously for a single patient.
“People anywhere in the world could print our design. You can envision a future where your doctor will have a 3-D printer and can print out the devices as needed,” said Luis Fernando Velásquez-García, a researcher in the Microsystems Technology Laboratories. “If someone has cancer, you can take a bit of tissue in our device, and keep the tumor alive, to run multiple tests in parallel and figure out what would work best with the patient’s biological makeup. And then implement that treatment in the patient.”
One potential application is testing immunotherapy, a new treatment method that uses drugs to “rev up” a patient’s immune system to help it fight cancer.
“Immunotherapy treatments have been specifically developed to target molecular markers found on the surface of cancer cells,” said graduate researcher Ashley Beckwith. “This helps to ensure that the treatment elicits an attack on the cancer directly while limiting negative impacts on healthy tissue. However, every individual’s cancer expresses a unique array of surface molecules — as such, it can be difficult to predict who will respond to which treatment. Our device uses the actual tissue of the person, so is a perfect fit for immunotherapy.”
The research was published in a paper entitled “Monolithic, 3D-Printed Microfluidic Platform for Recapitulation of Dynamic Tumor Microenvironments.”
“A key challenge in cancer research has been the development of tumor microenvironments that simulate mechanisms of cancer progression and the tumor-killing effects of novel therapeutics,” said Jeffrey T. Borenstein, who leads the immuno-oncology program at Draper. “Through this collaboration with Luis and the MTL, we are able to benefit from their great expertise in additive manufacturing technologies and materials science for extremely rapid design cycles in building and testing these systems.”
Microfluidic devices are typically produced via micromolding with PDMS. The technique was not suitable, however, for producing a device with fine 3D features such as the fluid channels, so the researchers turned to 3D printing, which allowed them to create the device in one piece. They experimented with several resins, but finally settled on Pro3dure GR-10, which is often used to make mouth guards. The resin is nearly as transparent as glass, can be printed in very high resolution, and has hardly any surface defects – and it doesn’t harm the cells.
“When you print some of these other resin materials, they emit chemicals that mess with cells and kill them. But this doesn’t do that,” Velasquez-Garcia said. “To the best of my knowledge, there’s no other printable material that comes close to this degree of inertness. It’s as if the material isn’t there.”
The device also features a “bubble trap” and a “tumor trap.” Fluids flowing into a device like this one creates bubbles that can disrupt the experiment or burst and release air that destroys tumor tissue. So the researchers created a bubble trap, a chimney that rises from the fluid channel into a threaded port through which air escapes. Fluid gets injected into an inlet port adjacent to the trap, then flows past the trap, where any bubbles in the fluid rise up through the threaded port and out of the device. Fluid is then routed around a small U-turn into the tumor’s chamber, where it flows through and around the tumor fragment.
The tumor trap sits at the intersection of the larger inlet channel and four smaller inlet channels. Tumor fragments, less than one millimeter across, are injected into the inlet channel via the bubble trap. As the fluid flows through the device, the tumor is guided downstream to the tumor trap, where it gets caught. The fluid continues traveling along the outlet channels, which are too small for the tumor to fit into, and drains out of the device. A continuous flow of fluids keeps the tumor fragment in place and constantly replenishes nutrients for the cells.
“Because our device is 3-D printed, we were able to make the geometries we wanted, in the materials we wanted, to achieve the performance we wanted, instead of compromising between what was designed and what could be implemented — which typically happens when using standard microfabrication,” Velásquez-García said.
The next step is to test how the tumor fragments respond to therapeutics.
“The traditional PDMS can’t make the structures you need for this in vitro environment that can keep tumor fragments alive for a considerable period of time,” said Roger Howe, a professor of electrical engineering at Stanford University, who was not involved in the research. “That you can now make very complex fluidic chambers that will allow more realistic environments for testing out various drugs on tumors quickly, and potentially in clinical settings, is a major contribution.”
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
[Source/Images: MIT]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Printing Money Episode 38: Additive Manufacturing Deal Analysis with Rajeev Kulkarni
Welcome to Printing Money Episode 38. Rajeev Kulkarni returns for this episode, and we find it hard to believe it’s been nearly two years since his first appearance. In the interim,...
3D Printing News Briefs, May 14, 2026: Project Calls, Reseller, Reconstructive Surgery, & More
We’re starting off today’s 3D Printing News Briefs with two new Project Calls from America Makes. We’ll move on to some more business, with Axtra3D expanding its presence in North...
Fabric8Labs & University of Illinois Collaborate on 3D Printed Copper Cold Plates for Data Centers
Collaboration between emerging technology enterprises and research universities is one of the most consistently winning tactics for any nation building (or rebuilding) an industrial ecosystem. It’s an especially constructive approach...
3D Printing News Briefs: May 7, 2026: Metal Powder Bed Fusion, Surgical Plates, & More
In today’s 3D Printing News Briefs, we’ll start with a strategic collaboration to advance next-generation metal additive manufacturing (AM), before moving on to funding for surgical research. We’ll end with...