Genetically modified organisms, or GMOs, are subject to strict regulation. In a paper entitled “An Additive Manufacturing Approach that Enables the Field Deployment of Synthetic Biosensors,” a group of researchers explains that living biosensors, which can be deployed for disease diagnosis in the field, are considered GMOs and are therefore subject to that same regulation. Using 3D printing is one way in which these biosensors can be made secure and acceptable for deployment in the field.
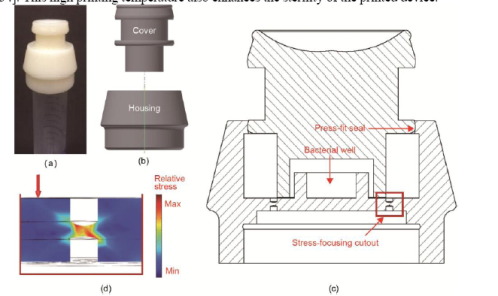
In rural and less-developed areas, the researchers explain, the pathogen P. aeruginosa can be acquired in local clinics. The organism is found in lung sputum samples upon infection, and the samples can be collected and placed in commercially available sample tubes. The researchers’ study focuses on creating a biosensor that could be used to immediately diagnose the disease in the field. They used E. coli as a biosensor, but needed a way to safely transport the dangerous bacteria from the laboratory. So they 3D printed a unique cap that completely encases the bacteria and can be securely sealed onto a sample tube containing lung sputum samples.
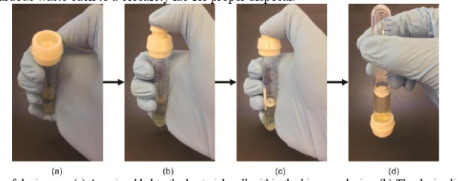
 When the sample is ready to be analyzed, force is applied with the user’s finger to the cap, which causes the E. coli biosensor to break out of its 3D printed housing and drop into the sample.
When the sample is ready to be analyzed, force is applied with the user’s finger to the cap, which causes the E. coli biosensor to break out of its 3D printed housing and drop into the sample.
“Detection of 3O-C12, which will be produced by P. aeruginosa in the sample or is already present in the sample, causes the living biosensor to produce mCherry and turns the sample red; this color can be detected through fluorescence analysis or visually with the human eye,” the researchers explain. “Thus, a colorimetric change in the sample can indicate the presence of P. aeruginosa in the lungs.”
P. aeruginosa, they continue, functions as an opportunistic pathogen in the lungs of patients with cystic fibrosis. Currently, it is diagnosed either with enzyme-linked immunosorbent assays (ELISAs) and real-time polymerase chain reaction (PCR) assays, which are either expensive or require extensive training to administer. The researchers’ hope is that their biosensor, with its secure 3D printed containment device, can be used as an inexpensive, easy-to-use alternative.
The 3D printed device was designed in Autodesk Inventor and 3D printed in ABS ULTRAT using a Zortrax M200 3D printer.
“To encase the 3O-C12 living biosensor into the 3D-printed device, a 40 µL agar plug was added to the bacterial well in the housing; the engineered E. coli cells were then plated onto the agar surface,” the researchers state. “After overnight culture, the plunger was pressed into the housing and a press-fit seal was formed to isolate the bacterial cells from the environment. The cap containing the living biosensor could then be safely transferred to the field and stored in a refrigerator for up to 30 d before use.”
The biosensor quickly senses the presence of the P. aeruginosa pathogen, and because of its enclosure within the 3D printed device, it should face far fewer regulatory hurdles, allowing it to be deployed freely outside the lab.
“The 3D-printed device turns the GMO into a component of the overall device and ensures that the biosensor is isolated from the environment,” the researchers conclude. “Thus, 3D printing this device enables our living biosensor, with its versatility to process environmental input, to be field-deployed as a novel, cost-effective diagnostic technology.”
Authors of the paper include Daniel Wolozny, John R. Lake, Paul G. Movizzo, Zhicheng Long and Warren C. Ruder.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
The Dental Additive Manufacturing Market Could Nearly Double by 2033, According to AM Research
According to an AM Research report from 2024, the medical device industry, specifically in dentistry, prosthetics, and audiology, is expected to see significant growth as these segments continue to benefit from...
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
AM Research Webinar Explores Continuum’s Sustainable Metal Additive Manufacturing Powders
Metal additive manufacturing (AM) powder supplier Continuum Powders is working to develop solutions that empower industries to reduce waste and optimize their resources. An independent life cycle assessment (LCA) of...
3D Printed Footwear Startup Koobz Lands $7.2M in Seed Round
California-based Koobz is focused on reshoring the U.S. footwear supply chain with advanced manufacturing processes, including 3D printing. The startup just announced that it has added $6 million to its...