In 3D bioprinting, scientists and researchers use bioinks for material, which are loaded with cells to 3D print biological structures; once printed, secondary crosslinking mechanisms come into play to retain the shape’s structural fidelity. These bioinks can facilitate cell adhesion, proliferation, and differentiation, and also exhibit all the characteristics of an extracellular matrix environment – making it possible to create patient-specific human tissues in a laboratory setting.
A lot of research goes into developing the best bioinks, and we’ve seen them made from materials like stem cells, sugarcane waste, and gelatin hydrogels, but according to a new collaborative study by researchers from the University of Texas at El Paso, the University of Texas at Austin, and RIKEN in Japan, most don’t have both incompatibility and high fidelity, which means they can’t replicate the same kind of complexity that’s found in biological organs and tissues.

Gross Morphology of lattice and rectangular-sheet structures printed using pluronic and gelatin. (AI,BI) depict the stl. file image for the lattice and rectangular structures, respectively. (AII,BII) represent the en-face images for the same structures printed using Pluronic F-127. (AIII,BIII) are representations of the en-face images for lattice and rectangular patterns printed using the f-gelatin-based bioink, respectively.
The team “optimized the properties of a visible light crosslinkable gelatin-based bioink” in a previous study, and were able to 3D print bilayer, rectangular sheet structures that were infused with STO fibroblasts and C2C12 myoblasts. But, they were not able to assess the ability of the bioink to create complex lattice structures, which can be used as physical scaffolds for cells to grow on.
So, in their most recent published paper, titled “A Comparative Study of a 3D Bioprinted Gelatin-Based Lattice and Rectangular-Sheet Structures,” the team focused on determining if it was possible to make a 3D bioprinted lattice structure with the same f-gelatin-based bioink, and then use factors such as porosity, rheological properties, and structural fidelity to compare it with their previous structure.
The abstract reads, “Current bioinks lack both high printability and biocompatibility required in this respect. Hence, the development of bioinks that exhibit both properties is needed. In our previous study, a furfuryl-gelatin-based bioink, crosslinkable by visible light, was used for creating mouse mesenchymal stem cell-laden structures with a high fidelity. In this study, lattice mesh geometries were printed in a comparative study to test against the properties of a traditional rectangular-sheet. After 3D printing and crosslinking, both structures were analysed for swelling and rheological properties, and their porosity was estimated using scanning electron microscopy. The results showed that the lattice structure was relatively more porous with enhanced rheological properties and exhibited a lower degradation rate compared to the rectangular-sheet. Further, the lattice allowed cells to proliferate to a greater extent compared to the rectangular-sheet, which initially retained a lower number of cells. All of these results collectively affirmed that the lattice poses as a superior scaffold design for tissue engineering applications.”
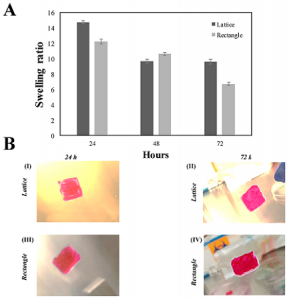
(A) Swelling analysis for both the f-gelatin-based lattice and rectangular-sheets over a period of three days after being subjected to visible light crosslinking. (B) Shown above in (BI,II) is cell-laden lattice constructs. In (BIII,IV) is cell-laden rectangular-sheet constructs. (BI,III) was acquired after 24 h of culture. (BII,IV) was acquired after 72 h of culture. No significant differences in the degradation rates of both structures were evident at 72 h of incubation.
The team used an Allevi 2 3D bioprinter, previously known as the BIOBOT 1, to create the cell mixture and bioink that they’d optimized in the previous study. A mixture of RB, HA, and f-gelatin were used to make the bioink. They believed that the experiments they performed for the study would help them see more differences between the rectangle and lattice structures, as well as “open up the possibility of significantly enhancing the design of a 3D bioprinted construct for engineering cardiac tissue-on-a-chip, using bioprinting.”
The researchers determined that, when compared to the rectangular-sheet structure, the multi-layered lattice structure appeared to be more the more stable scaffold, and provided optimal porosity, well-connected pores, and more structural rigidity, all of which combined so that the encapsulated cells would proliferate more. This means that lattice structures are likely a better choice for a scaffold for 3D bioprinted structures in tissue engineering applications.
“This study revealed fundamental differences in two dissimilar, chemically cross-linked soft structures fabricated using the same bioink,” the researchers wrote. “This fundamental difference gave rise to divergent mechanical properties of both structures, which may have also influenced cell behavior, growth, and proliferation, enabling the lattice structure to allow cells to communicate better, eventually resulting in better cell yields compared to the rectangular-sheet. In addition, the lattice allows more bioink volume to be deposited, thereby allowing a greater number of cells to be encapsulated within the printed structure.”
They concluded that in the future, lattice structures in 3D bioinks can be investigated as good platforms for culturing a variety of cell types, in a layer-by-layer fashion, in the same structure.

A representative SEM cross-section image of a gelatin lattice structure that was acquired in order to determine the apparent porosity and average pore size (A). The cross-sectional SEM image for the rectangular-sheet structure printed using f-gelatin was previously reported (B).
Co-authors of the paper are Shweta Anil Kumar, Nishat Tasnim, Erick Dominguez, Shane Allen, Laura J. Suggs, Yoshihiro Ito, and Binata Joddar.
Discuss this research and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
Nylon 3D Printed Parts Made More Functional with Coatings & Colors
Parts 3D printed from polyamide (PA, Nylon) 12 using powder bed fusion (PBF) are a mainstay in the additive manufacturing (AM) industry. While post-finishing processes have improved the porosity of...
$25M to Back Sintavia’s Largest Expansion of Metal 3D Printing Capacity Since 2019
Sintavia, the digital manufacturing company specializing in mission-critical parts for strategic sectors, announced a $25 million investment to increase its production capacity, the largest expansion to its operations since 2019....
Velo3D Initiates Public Offering in a Bid to Strengthen Financial Foundations and Drive Future Growth
Velo3D (NYSE: VLD) has been among a number of publicly traded 3D printing firms that have attempted to weather the current macroeconomic climate. After posting a challenging financial report for 2023,...