Researchers Test 3D Printed Lattice Structures as Implants to Facilitate Bone Growth
 The advances being made in bioprinting and tissue engineering mean that even the most severe injuries have a chance at being repaired. Loss of bone due to injury or disease used to be a permanent condition, but that’s not the case anymore – medical technologies including 3D printing are now capable of replacing or even restoring lost bone tissue. New tissue can actually be grown from scaffolds that are implanted inside the body, as long as those scaffolds are made from osteoconductive material, which is a material that is capable of stimulating the growth of bone tissue.
The advances being made in bioprinting and tissue engineering mean that even the most severe injuries have a chance at being repaired. Loss of bone due to injury or disease used to be a permanent condition, but that’s not the case anymore – medical technologies including 3D printing are now capable of replacing or even restoring lost bone tissue. New tissue can actually be grown from scaffolds that are implanted inside the body, as long as those scaffolds are made from osteoconductive material, which is a material that is capable of stimulating the growth of bone tissue.
A group of researchers recently conducted a study in which they used selective laser melting (SLM) 3D printing to generate titanium lattice structures as scaffolds for bone regeneration. The research is published in a paper entitled “Osteoconductive Lattice Microarchitecture for Optimized Bone Regeneration,” which you can access here.
“Bone replacement materials need to be accepted by the body and should clinically be infiltrated with bone tissue within a short time, so ideally they are osteoconductive,” the researchers explain. “Bone tissue engineering of osteoconductive biomaterials like other tissue engineering approaches normally relies on the combination of cells, bioactive factors, and biomaterial scaffold to facilitate and accelerate the regeneration of bone tissue.”
The shape of the bone scaffold can be designed to perfectly match the size and shape of the bone defect. In the study, the researchers 3D printed titanium structures to replace bone in rabbits, using SOLIDWORKS software to design the implants.
“The outer macrogeometry of the implants is designed as stepped cylinders of 7.5 mm, respectively, 6 mm diameter and a height of 4.2 mm,” the researchers state. “The inner microarchitecture is constructed by cutting out symmetrically arranged square channels in all three orthogonal directions, see Figure 1. Based on most promising scaffold parameters from the literature, the width of the channels w (distance between the rods) and the wall thickness s (rod caliber) of the remaining trusses have been systematically varied in a way that complete layers of cubic unit cells are formed along the cylindrical main axis (see Fig. 1 for an overview of the designs and Table 1 for the structural values). Finally, a 0.2 mm fine ring completes the outermost margin of the framework to close the last layer and prevent sharp edges during surgery.”
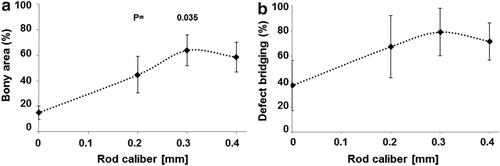
The implants were 3D printed using a Realizer 250HT from SLM Solutions. 18 adult rabbits were used in the study; they were housed in groups of two to four and each had four scaffolds applied to them at random. After four weeks, the implants were removed and studied to see how well they had stimulated bone growth, with distinctions being made between implants with different rod distance and rod caliber in a lattice titanium microarchitecture. The results showed that the best results were achieved at a rod distance of 0.8 mm and a rod caliber of 0.3 to 0.4 mm.
“Since AM and 3DP in many cases yield in a lattice microarchitecture, these results suggest to overcome the former dogma on channels of 0.3 to 0.5 mm and substitute them by channels of 0.8 mm for bone substitutes, where bone ingrowth has to occur fast and efficiently,” the researchers conclude.
Authors of the paper include Michael De Wild, Chafik Ghayor, Simon Zimmerman, Jasmine Rüegg, Flora Nicholls, Felix Schuler, Tse-Hsiang Chen, and Franz Z. Weber.
Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: Velo3D Revenue Up Fueled by Defense Momentum
Velo3D (Nasdaq: VELO) reported a strong start to 2026, with revenue rising as defense and aerospace customers continued shifting from pilot programs into full-scale additive manufacturing (AM) production. The company...
AMS 2027: The AM Industry’s Biggest Business Conversations Return to New York
Additive Manufacturing Strategies (AMS) is heading back to New York next February for what has become one of the industry’s most important business gatherings. The 10th annual AMS conference (or...
AM & the Military’s Self-Infliction of Rapid Change
I’ve noted before that the additive manufacturing (AM) market for defense has started to evolve so quickly that it’s impossible to even keep track of all the updates in real...
ROBOZE Buys Dimanex Assets to Build “Physical AI” Platform
Dutch firm Dimanex got its start as an MRO platform for the railways. The company had a contract with the Dutch Army in 2018, and later that year signed one...