Connecticut: A very exciting collaboration is in the works to benefit biomedical applications powered by 3D printing.
Connecticut: A very exciting collaboration is in the works to benefit biomedical applications powered by 3D printing.
Oxford Performance Materials hits the headlines again, and this time they are teaming up with their Ivy League Connecticut neighbors at Yale University to do some heavy-hitting research in exploring biomedical applications with 3D printing — and OPM’s high performance polymer, poly-ether-ketone-ketone (PEKK).
 This research venture between OPM and Yale will comprise ten projects with seven members of the Yale faculty working to delve further into what has become a speciality for OPM — 3D printing applications for biomedical devices. The teams will be working to develop:
This research venture between OPM and Yale will comprise ten projects with seven members of the Yale faculty working to delve further into what has become a speciality for OPM — 3D printing applications for biomedical devices. The teams will be working to develop:
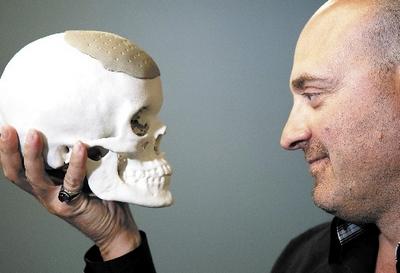
- New PEKK-based cranial and facial devices that support direct tissue attachment.
- 3D printed PEKK prosthesis for rib replacement.
- 3D printed PEKK devices that deliver therapeutics for improved vertebral fusion.
- Devices that deliver antibiotics to combat the pressing burden of implant associated infections.
“We are excited to announce this broad collaboration with Yale’s distinguished team of biomedical engineering and clinical experts,” said Dr. Adam Hacking, PhD, Chief Scientific Officer, Oxford Performance Materials. “Our comprehensive medical device development platform will support the innovation and dedication to improving patient care for which Yale and its faculty are recognized.”
Together, we foresee the development and delivery of new medical technologies and applications in ways that were previously not possible. OPM has made significant progress with our PEKK-based platform, and we are continuing to advance this technology to improve patient outcomes with our partners at Yale.”
Founded in 2000, OPM has made a name for themselves with the 3D printing of cranial and facial implants. OPM is a high performance plastics company, and their secret to success is centered around materials built using the PEKK molecule. OPM develops advanced materials technology for the areas of 3D printed biomedical devices, biomedical raw materials and 3D printed industrial parts.
“PEKK is biocompatible, radiolucent, strong and durable. 3D printing enables the facile fabrication of complex geometries. In combination, these technologies present new and previously unavailable opportunities to develop and deliver customized, patient-specific therapies that are also economically viable,” said Professor Mark Saltzman, PhD, Yale’s Goizueta Foundation Professor of Chemical and Biomedical Engineering and Chair of Biomedical Engineering. “We are looking forward to working with Dr. Hacking and his team at OPM on these joint research initiatives that are designed to improve how personalized medicine is developed and delivered to patients.”
 The combination of a renowned university with a rich history in medical research working with the dynamic innovative momentum of OPM is a perfect match, not to mention a substantial collaboration which should yield results with impact in the area of biomedical research.
The combination of a renowned university with a rich history in medical research working with the dynamic innovative momentum of OPM is a perfect match, not to mention a substantial collaboration which should yield results with impact in the area of biomedical research.
“This joint research program between Yale University and Oxford Performance Materials is a prime example of the types of collaborations that we are working to develop and nurture here in Connecticut,” said Connecticut Governor Dannel P. Malloy. “Our state has long been an incubator for scientific, technological and medical innovation, and this program has the potential to yield significant benefits to society through 3D printing, advanced materials science and strategic partnership between private industry and academia. This is how innovation is fostered in the 21st century economy, and we are proud to support both Yale and OPM as they embark on this groundbreaking research right here in Connecticut.”
What do you think of the projects these two entities are working on? Discuss the implications of 3D printing and biomedical devices with us in the Yale University and OPM Collaborate forum at 3DPB.com.
Special notes on OPM:
- They are the only company to receive FDA clearance to manufacture 3D printed polymeric implants for its PEKK cranial devices.
- OPM received a second FDA clearance for its patient-specific facial implants in July 2014.
- OPM was also the recipient of the New Economy “Most Innovative Company in 3D Healthcare Technology” Award in 2014.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Gorilla Sports GE’s First 3D Printed Titanium Cast
How do you help a gorilla with a broken arm? Sounds like the start of a bad joke a zookeeper might tell, but it’s an actual dilemma recently faced by...
Nylon 3D Printed Parts Made More Functional with Coatings & Colors
Parts 3D printed from polyamide (PA, Nylon) 12 using powder bed fusion (PBF) are a mainstay in the additive manufacturing (AM) industry. While post-finishing processes have improved the porosity of...
$25M to Back Sintavia’s Largest Expansion of Metal 3D Printing Capacity Since 2019
Sintavia, the digital manufacturing company specializing in mission-critical parts for strategic sectors, announced a $25 million investment to increase its production capacity, the largest expansion to its operations since 2019....
Velo3D Initiates Public Offering in a Bid to Strengthen Financial Foundations and Drive Future Growth
Velo3D (NYSE: VLD) has been among a number of publicly traded 3D printing firms that have attempted to weather the current macroeconomic climate. After posting a challenging financial report for 2023,...