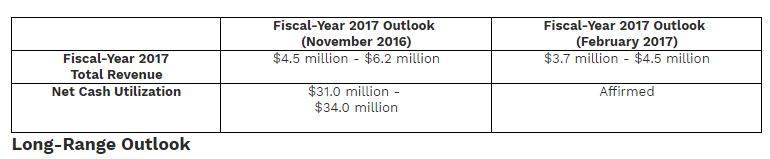Organovo Releases Fiscal Report for Q3 2017; Offers Projections for the Year
 As Organovo leads in the realm of 3D printed human tissue, it’s no surprise to see that revenues are up once again—and substantially so. Reporting fiscal results for Q3 2017 and updating their outlook for the year, Organovo does, however, show a net loss of $9.6 million, or $0.09 per share—down from $10.5 million, or $0.11 per share, for the fiscal third quarter of 2016.
As Organovo leads in the realm of 3D printed human tissue, it’s no surprise to see that revenues are up once again—and substantially so. Reporting fiscal results for Q3 2017 and updating their outlook for the year, Organovo does, however, show a net loss of $9.6 million, or $0.09 per share—down from $10.5 million, or $0.11 per share, for the fiscal third quarter of 2016.
“We grew total revenue at a strong year-over-year pace during the fiscal third quarter, showing continued uptake of our tissue research services,” said Keith Murphy, CEO, Organovo. “We recognized revenue from six new customers and seven repeat customers during the period, demonstrating market penetration and solid repeat business. We also added one global Top 25 pharma customer to our roster, bringing our total to eleven with this key group.”
Highlights for Fiscal Third-Quarter 2017 are as follows:
- Revenues for products and services were $0.7 million, showing a whopping increase of 139% in comparison to this time last year. Organovo states this is due to contracts for tissue research. Collaboration and grants brought in revenue of $0.4 million, thanks to the company’s ongoing collaboration with Merck, one of the largest pharmaceutical companies in the world.
- Operating expenses showed cost of revenues at $0.2 million, R&D with an increase of 10% at $5 million, and selling, general, and administrative costs at $5.5 million—down 11% from this time last year.
- For Liquidity and Capital Resources, Organovo ended the quarter with a cash and cash equivalents balance of $70.0 million. Working capital was $67.5 million, in comparison to $65.2 million for this quarter last year.
Organovo’s 2017 projections for the year look toward total revenues of between $3.7 million and $4.5 million, in comparison to 2016 at $1.5 million. Net cash utilization is projected between $31.0 million and $34.0 million. They expect both the ExVive Human Liver Tissue and ExVive Human Kidney Tissue services to grow by tens of millions, and into addressable markets of $1 billion and $2 billion, respectively.
“We’re revising our total revenue guidance for fiscal 2017 because of a change in the timing of customer orders due to specific customer requests for additional validation studies related to certain use cases and for qualification of an additional cell source. We expect to successfully complete the additional scientific studies required to address these issues, and don’t anticipate these items will have a long-term impact on customer adoption. The required studies will delay a portion of our forecasted revenue into fiscal 2018. We’re working diligently with our customers to complete the technical work and to unlock both existing backlog and prospective orders, and look ahead with confidence to continued growth in our pipeline. In addition, our internal liver validation data now includes successful identification of toxicity for two out of three proprietary compounds that were classic preclinical misses for one of our Top 10 pharma customers. This hit rate is consistent with our overall testing success and further demonstrates significantly increased predictive power for our customers,” said Murphy.
“During the fiscal third quarter, we also announced our 3D bioprinted human liver as the first candidate in our therapeutic tissues portfolio and presented early data showing survival and sustained functionality of this tissue when implanted into animal models at the Tissue Engineering & Regenerative Medicine International Society Conference (TERMIS). We expect to optimize the final tissue design and are embarking on pre-GLP safety and efficacy studies that will take us through the next 18 months. Based on our path forward, we now expect to target an IND submission with the liver tissue during the calendar year 2020.”
Headquartered in San Diego, California, Organovo’s work is undeniably important on numerous levels as they work with researchers, pharmaceutical companies, and establish other collaborative endeavors in regenerative medicine. As their progress in building with cells through 3D printing continues steadily, the scientific world looks to them for accelerated research in medical treatments, to include toxicology studies and preclinical drug testing. Recently, they have produced exclusive findings regarding their 3D printed liver tissue, as well as embarking on new research regarding bioprinted kidney tissue.
‘Tissue on demand’ is the ultimate goal of this futuristic company—resting on a solid foundation due to the strength of their investors. Discuss in the Organovo forum at 3DPB.com.
[Source: Organovo]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing News Briefs, December 31, 2025: Frenzy Engine, Adaptive Slicing, & More
It’s the last 3D Printing News Briefs of the year! On this New Year’s Eve, we’re focusing on Beehive’s completed Frenzy Engine, adaptive slicing for binder jetting, medical 3D printing...
Disney Accelerator Backs Large-Format Robotic 3D Printing Service Haddy
A few months ago, Haddy, a contract manufacturer based in Florida that leverages robotic arm additive manufacturing (AM) systems to produce furniture, announced that it had opened what it was...
Disney’s 3D Printed Robot Steals the Show at IEEE IROS 2023 Conference
At the intersection of imagination and cutting-edge technology, Disney Research has outdone itself once again. Using 3D printing, they unveiled a captivating child-size bipedal robot at the 2023 IEEE/RSJ International...
3D Printing Bring Disney’s “Elemental” to Life for Film’s Premier
As Disney and Pixar’s latest animated feature Elemental debuts today, the field of movie premieres experiences a technological transformation. Remarkably, the film’s characters have already made appearances at some of...