New Frozen-Form Additive Manufacturing System Holds Potential for Better 3D Printed Tissue Scaffolds
 While the many forms of 3D printing live tissue are generally grouped together under the general category of 3D bioprinting, the methods are so varied that some of them barely resemble each other. The entire concept of bioprinting is still hard to wrap one’s mind around, yet scientists are constantly coming up with new variations of the technology.
While the many forms of 3D printing live tissue are generally grouped together under the general category of 3D bioprinting, the methods are so varied that some of them barely resemble each other. The entire concept of bioprinting is still hard to wrap one’s mind around, yet scientists are constantly coming up with new variations of the technology.
One of the major varying factors in the technology is how scaffolds are created. The scaffold, which serves to give structure to the printed cells as they develop and grow, is a key component of the bioprinting process, and how the scaffold is created can make the difference between success or failure. A group of researchers in Taiwan have developed a new method of fabricating scaffolds using a technique known as the frozen-form method, or FFM, also known as low-temperature deposition manufacturing.
In a paper entitled “Design and Development of a Novel Frozen-Form Additive Manufacturing System for Tissue Engineering Applications,” the researchers explain why additive manufacturing has become an increasingly popular method of fabricating scaffolds – namely, because the technology allows scientists to have precise control over the size, shape and porosity of the scaffold, engineering it to fit the needs of a specific patient.
However, the most common methods of 3D printing tissue scaffolds – SLS, SLA and FDM – each have their drawbacks, the researchers point out. SLS is limited in terms of the materials that can be used, while with SLA, the photoinitiators in the resin materials can leave residues or byproducts that have cytotoxic effects on the cells embedded in the scaffolds. Scaffolds created with FDM technology have excellent mechanical properties, but they require high temperatures and tend to shrink and distort when they cool. In addition:
“In our previous FDM research, PLA22 and polylactic-co-glycolic acid (PLGA) have been used as building materials,” the researchers state. “We discovered that the average molecular weight of PLGA decreased to ~22.4% of its original value after the FDM process. This occurred because thermal hydrolysis occurs during the melt extrusion process, which tends to reduce the structural strength of the scaffolds. As with SLS, the operating temperature of FDM is excessively high for biomolecules in terms of growth factors. Therefore, incorporating biomolecules into the scaffold is difficult.”
 To circumvent these issues, what the research team calls a “pseudo-FDM” technique, called low-temperature deposition modeling (LDM), was developed.
To circumvent these issues, what the research team calls a “pseudo-FDM” technique, called low-temperature deposition modeling (LDM), was developed.
“The principle of LDM is similar to that of FDM, except that a low-temperature cooling platform is required and the material-feeding mechanism employs a syringe or screw rather than an extruder,” the study continues. “LDM deposits polymer that has been dissolved in a solvent into the low-temperature cooling platform. The temperature of this platform is far below the crystallization temperature (i.e., freezing point) of the solvent. Therefore, the polymer solution freezes rapidly, and a thermally induced phase separation process occurs. After postprocess freeze-drying has been applied, the solvent is extracted, and the polymer remains as the scaffold.”
The advantages of LDM are many: scaffolds created with the technology can be both macroporous and microporous, and biomolecules or bioactive compounds can be incorporated during the build process. The polymer’s molecular weight is barely affected by the process, and the technology is suitable for multiple synthetic or natural macromolecular materials such as chitosan, alginate, PLA, polyglycolic acid (PGA), PCL, PLGA, and polyurethane. The LDM process involves the transition of a liquid into a solid, which is why it’s also known as a frozen-form method (FFM), or, for the purpose of this study, a frozen-form additive manufacturing system (FFAMS).
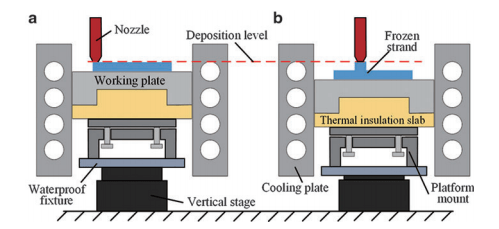
There are several different methods of creating a low-temperature environment for the FFM process, but they each have their drawbacks, as well. Therefore, the research team decided to create a new method of creating a low-temperature environment, one that would result in a compact and practical FFAM machine. To do so, they first designed a uniform cryogenic device (UCD) that utilizes four cooling plates to create an enclosure, or “cooling area.” A working plate, designed so that it can be moved up and down inside the cooling area, was placed inside as a build plate onto which material could be deposited. After each layer is deposited, the plate was lowered so that the next layer could be deposited at the exact same height as the previous layer, thus ensuring a consistent temperature.
The UCD was only one module of the system. The material dispenser module was composed of an air-powered fluid dispenser, which acted as an extruder into which the researchers placed a mixture of PCL-based waterborne polyurethane. They then experimented by printing scaffolds with the material, measuring temperature as each layer was deposited, as well determining the optimal nozzle diameter and layer thickness for different scaffold shapes. Rather than using a Cartesian system as most FFM methods do, they used a SCARA robot for the motion module, which gave them more flexibility.
“In our study, the low-temperature cooling area was designed to fit inside a small space so that sufficient room surrounding the SCARA remained for the construction of various types of work stations, such as a cell-seeding platform,” the team states. “Therefore, the FFAMS could be expanded as a multifunctional system or a continuous production line for scaffold production in the interest of commercialization.”
 You can read the details of the experiments and their results in the full study, which you can download here. Overall, the researchers concluded, their study produced flexible scaffolds with high potential for future use in tissue engineering and soft tissue repair. While the study focused on the production of scaffolds with simple shapes like tubes and squares, they intend to continue their research in the future to develop complex scaffolds that mimic biological systems, as well as larger, taller scaffolds for the creation of cartilage tissue for the nose, ear and trachea.
You can read the details of the experiments and their results in the full study, which you can download here. Overall, the researchers concluded, their study produced flexible scaffolds with high potential for future use in tissue engineering and soft tissue repair. While the study focused on the production of scaffolds with simple shapes like tubes and squares, they intend to continue their research in the future to develop complex scaffolds that mimic biological systems, as well as larger, taller scaffolds for the creation of cartilage tissue for the nose, ear and trachea.
The study’s authors include Chau-Yaug Liao, Wei-Jen Wu, and Ching-Shiow Tseng of the Department of Mechanical Engineering at National Central University; Cheng-Tien Hsieh and Shan-hui Hsu of the Institute of Polymer Science and Engineering at National Taiwan University; and Niann-Tzyy Dai of the Division of Plastic and Reconstructive Surgery, Department of Surgery, Tri-Service General Hospital at the National Defense Medical Center. Discuss in the Frozen-Form AM forum at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
Precision at the Microscale: UK Researchers Advance Medical Devices with BMF’s 3D Printing Tech
University of Nottingham researchers are using Boston Micro Fabrication‘s (BMF) 3D printing technology to develop medical devices that improve compatibility with human tissue. Funded by a UK grant, this project...
3D Printing Webinar and Event Roundup: April 21, 2024
It’s another busy week of webinars and events, starting with Hannover Messe in Germany and continuing with Metalcasting Congress, Chinaplas, TechBlick’s Innovation Festival, and more. Stratasys continues its advanced training...
3D Printing Webinar and Event Roundup: March 17, 2024
It’s another busy week of webinars and events, including SALMED 2024 and AM Forum in Berlin. Stratasys continues its in-person training and is offering two webinars, ASTM is holding a...
3D Printed Micro Antenna is 15% Smaller and 6X Lighter
Horizon Microtechnologies has achieved success in creating a high-frequency D-Band horn antenna through micro 3D printing. However, this achievement did not rely solely on 3D printing; it involved a combination...