Australia: Titanium Medical Implants Cure Patient Ills, But Stress the Wallet as Government Ignores Rebate Issues
 While many patients experiencing chronic pain around the world are very open to the idea of using 3D printing technology in medicine—as are their surgeons—some large and powerful entities like governments and insurance agencies may be responding with this: sorry, not so much. And while that’s frustrating to innovators, medical professionals, and the patients themselves, when it comes to rebates, reimbursement, and formal coverage, often nothing gets done without a long and delaying list of studies, considerations of liability, and number crunching—even when all of this comes on the heels of grants, support funds, and millions being given to programs that certainly seem related to new technology like 3D printing.
While many patients experiencing chronic pain around the world are very open to the idea of using 3D printing technology in medicine—as are their surgeons—some large and powerful entities like governments and insurance agencies may be responding with this: sorry, not so much. And while that’s frustrating to innovators, medical professionals, and the patients themselves, when it comes to rebates, reimbursement, and formal coverage, often nothing gets done without a long and delaying list of studies, considerations of liability, and number crunching—even when all of this comes on the heels of grants, support funds, and millions being given to programs that certainly seem related to new technology like 3D printing.
In the US, a lack of recognition by insurance companies for products like 3D printed implants can make numerous surgeries cost-prohibitive. While many of them are performed anyway due to life-threatening conditions, a lack of support in that area makes using the technology much more challenging, and surgeons are less inclined to use products that could in the end offer their patients greater comfort. An ongoing, related problem has shown itself too for Melbourne neurosurgeon Paul D’Urso in the area of titanium medical implants.
While the Australian government is talking a good technological talk up front, D’Urso contends that they aren’t walking the walk at all when it comes to giving rebates from private health funds. Not only could this be responsible for a possible stall-out in the progress of 3D printing in the medical arena, the financial obstacles are putting financial stress on Dr. D’Urso’s business, and patients.
All of this might be easier to swallow if D’Urso had not previously been highlighted by the Prime Minister as a pioneer in the field of making 3D printed skull and bone implants. He was lauded for his innovation last December as the government announced their $1 billion innovation and science agenda at CSIRO’s Discovery Centre. Now, while the enthusiasm seems to be there in terms of publicity and allocated broad-based funds, the finances are not coming together when the situation gets real in terms of patients and doctors needing support for the use of 3D printed implants.
Fortunately D’Urso does have some predominance and a voice within his community—and country—to speak out with, garnering some attention to the cause. And while of course his business interests are at stake here with Anatomics, a company he founded 20 years ago for creating customized 3D printed cranial implants, the larger issue is that the progress of using customized implants is being squelched, and ultimately, discouraged. Assuming no one wants to go backward at this point, D’Urso is now fighting for reimbursements to be given for custom 3D printed implants.
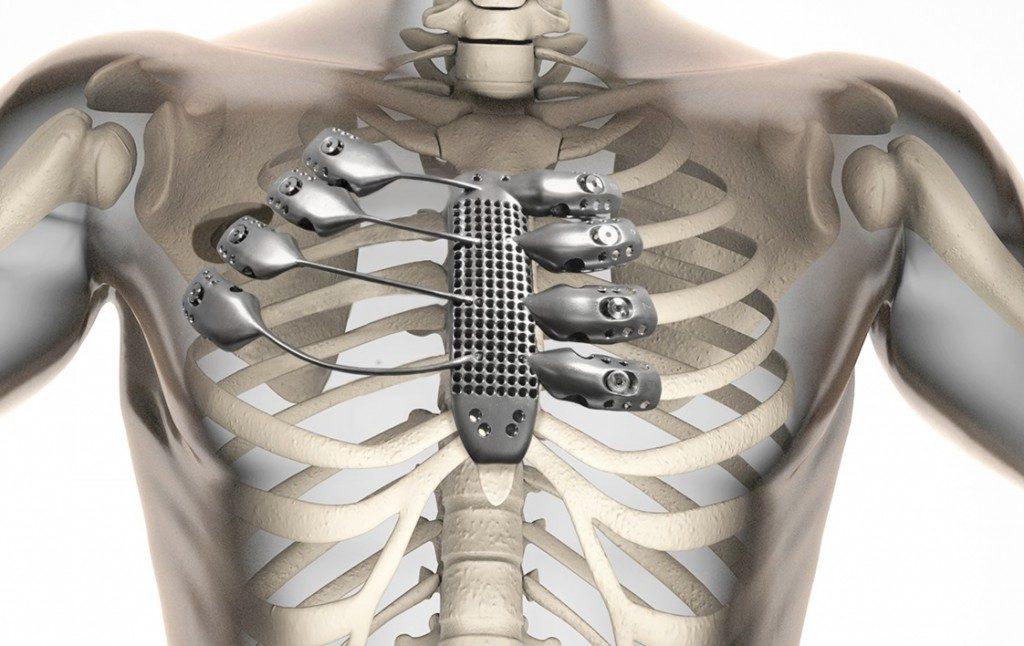
Anatomics is a leader in making implants for the spine, heel, and ribcage—as well as the hips and shoulders. Using progressive, strong new materials such as titanium, Anatomics is now exporting their implants to 30 different countries, as well as distributing them in Germany through B. Braun. The company also helped create a custom sternum and ribcage in a case that received quite a lot of attention last year. We followed this story as Anatomics was central in working with a medical team from Spain at the University of Salamanca. There, the team collaborated on a 3D printed rib and sternum implant, resulting in successful surgery and a patient thriving afterward.
The positive elements of the work being done by D’Urso and Anatomics are well-evidenced, but to promote the use of such implants further, health funds absolutely need to be reimbursed for the custom implants. Because they are not regulated by the government, this is not a technical obligation—nor is it being upheld, obviously.D’Urso currently must be dedicated to working on a case-by-case basis to get monies back from each patient’s case. Obviously this system needs to change, as up to eight weeks can be spent in negotiating funds—time which would be much better spent with those who are ill or for D’Urso to be in the lab innovating and creating other new medical solutions. The financial aspects are stressful for patients too, and in the end, more doctors decide to choose off-the-shelf, more basic implants that will receive automatic rebates, rather dealing with a hassle that seems to include everyone involved.

Prime Minister Malcolm Turnbull and then Minister for Industry, Innovation and Science Science Christopher Pyne – at the announcement being made in December at CSIRO. [Photo: Alex Ellinghausen]
“The patent is going ‘I’m in terrible pain’ and the doctor goes, ‘Ok, I’ll use the off-the-shelf product.’ We want to be regulated and then our business can accelerate and we can grow into a much bigger company and we can export it globally,” said D’Urso. “Our biggest hurdles are often just 20th century regulatory systems that are not allowing new technologies to emerge.”
CSIRO has offered great support via their manufacturing division as Anatomics has worked further in making titanium ink and developing 3D printing processes, such as those necessary to make the aforementioned 3D printed ribcage and sternum implant.
D’Urso points out the basic problem in that the government has the intention but they are showing an inability to follow through when it is crucial, and when the products being made locally are much better than generic ‘off-the-shelf’ implants. With customized implants being made within the country, patients in Australia also experience significant savings in import taxes.
“We are good at sending money and doing research but as soon as you put your head above the trenches and want to commercialize it, nobody knows what to do.”
Discuss further over in the Australian 3D Printed Titanium Implants forum at 3DPB.com.
[Source: AFR Weekend]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Inside The Barnes Global Advisors’ Vision for a Stronger AM Ecosystem
As additive manufacturing (AM) continues to revolutionize the industrial landscape, Pittsburgh-based consultancy The Barnes Global Advisors (TBGA) is helping shape what that future looks like. As the largest independent AM...
Ruggedized: How USMC Innovation Officer Matt Pine Navigates 3D Printing in the Military
Disclaimer: Matt Pine’s views are not the views of the Department of Defense nor the U.S. Marine Corps Throughout this decade thus far, the military’s adoption of additive manufacturing (AM)...
U.S. Congress Calls Out 3D Printing in Proposal for Commercial Reserve Manufacturing Network
Last week, the U.S. House of Representatives’ Appropriations Committee moved the FY 2026 defense bill forward to the House floor. Included in the legislation is a $131 million proposal for...