Last year, 3D Systems made big news when they shelled out $97 million to purchase software company Cimatron. The deal gave 3D Systems control of Cimatron’s major software product lines: CimatronE, CAD/CAM software for mold-, die- and tool-makers, and GibbsCAM, CAM software for production machining. Since the purchase, 3D Systems has been working on improving the software and making it their own, and now the company has announced the release of Cimatron 13, which contains a host of new and improved features that promise to increase productivity and minimize overhead in the manufacturing and design of molds, dies and discrete parts.
Last year, 3D Systems made big news when they shelled out $97 million to purchase software company Cimatron. The deal gave 3D Systems control of Cimatron’s major software product lines: CimatronE, CAD/CAM software for mold-, die- and tool-makers, and GibbsCAM, CAM software for production machining. Since the purchase, 3D Systems has been working on improving the software and making it their own, and now the company has announced the release of Cimatron 13, which contains a host of new and improved features that promise to increase productivity and minimize overhead in the manufacturing and design of molds, dies and discrete parts.
The software’s interface has been completely redesigned to make it faster and easier to learn and customize, which in itself is a major improvement, but there’s plenty more. Additional improvements include:
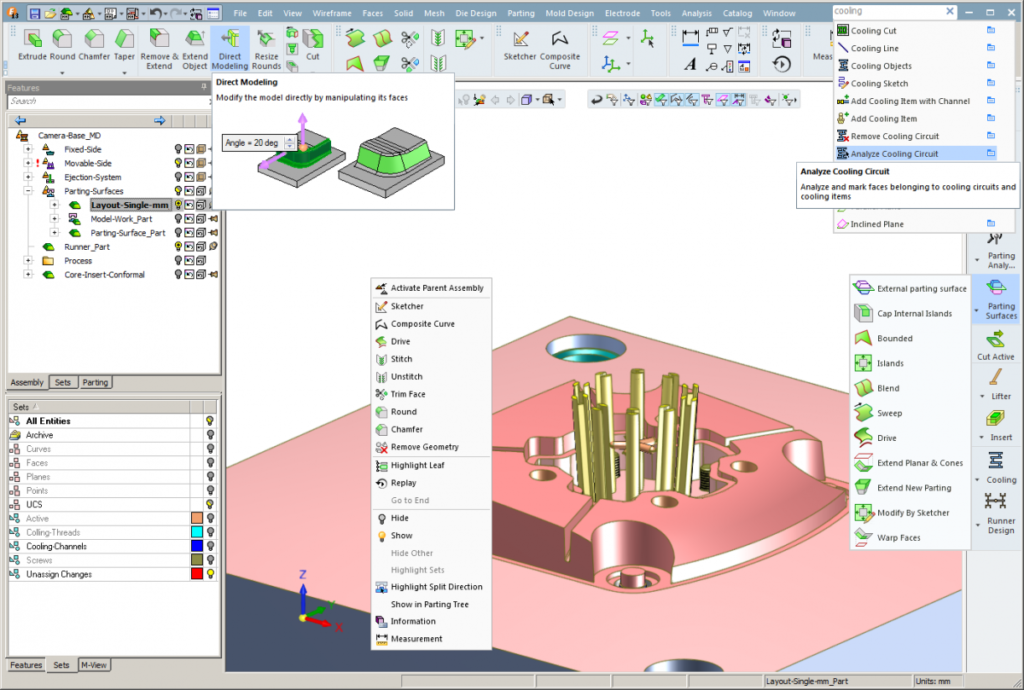
 Several new CAD for tooling functionalities that include direct modeling, enhanced assembly functions and new mesh operations with hybrid modeling capabilities
Several new CAD for tooling functionalities that include direct modeling, enhanced assembly functions and new mesh operations with hybrid modeling capabilities- Increased drafting capabilities, including the ability to create multiple views and shaded views
- New mold-design applicative tools such as gates design for a more streamlined process and conformal cooling tools to simplify the design of curved cooling channels
- Separate environments for progressive- and transfer-dies
- Smart electrode mirroring tools and hybrid design environments for faster electrode design with automated solid tools
- New plate machining system for efficient, automated programming of mold- and die-plates
- New measurement on CNC machines to define the measurement probing cycle in the NC environment, allowing validation of the machining process while the part is on the machine
- New and improved NC programming capabilities for milling and drilling, boosting programming automation in rough, finish and 2.5 axis operations
- Concurrent design and manufacturing environment that allows multiple users to work on the same project at the same time
“Cimatron 13 eases the growing pressure tool shops are facing to stay competitive in quality and delivery time,” said Calvin Hur, Vice President, Co-Chief Operating Officer & Chief Revenue Officer, Software Products, 3D Systems. “This significant update provides the means for tool shops to create better tools and parts, faster.”
One of Cimatron 13’s beta testers was Dayton, Ohio-based Bastech, a provider of additive manufacturing equipment and services. The company integrated Cimatron 13 into their workflow, speeding up the design of conformally-cooled injection molds, which were then printed on 3D Systems’ ProX DMP 200 metal 3D printer. Final inspection of the printed molds was carried out with Geomagic Control software. The whole process, of which Cimatron 13 was an integral part, drastically cut costs, design time, and manufacturing time.
“By using Cimatron to design conformal cooling, our process has changed from a very manual, to a very automated approach, saving us 70% on mold design time and cutting our mold costs by 16%, which led to a 14% reduction in injection cycle times and an overall increase in part quality,” said Ben Staub, President of Bastech. “Cimatron helps our mold-makers make better decisions about how to set up cavities, cores and inserts, before bringing them into reality using 3D Systems’ direct metal 3D printing.”
You can learn more about Cimatron 13 here. Discuss further in the Cimatron 13 software forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Unpeeled: New Arkema Material for HP, Saddle and Macro MEMS
A new Arkema material for MJF is said to reduce costs per part by up to 25% and have an 85% reusability ratio. HP 3D HR PA 12 S has been...
3D Printing News Briefs, January 20, 2024: FDM, LPBF, Underwater 3D Printer, Racing, & More
We’re starting off with a process certification in today’s 3D Printing News Briefs, and then moving on to research about solute trapping, laser powder bed fusion, and then moving on...
3D Printing Webinar and Event Roundup: December 3, 2023
We’ve got plenty of events and webinars coming up for you this week! Quickparts is having a Manufacturing Roadshow, America Makes is holding a Member Town Hall, Stratafest makes two...
Formnext 2023 Day Three: Slam Dunk
I’m high—high on trade show. I’ve met numerous new faces and reconnected with old friends, creating an absolutely wonderful atmosphere. The excitement is palpable over several emerging developments. The high...