Last week TriFusion Devices, an up and coming 3D printing company launched by three Texas A&M students, added another startup competition win to their rapidly growing pot of seed money. The company took home the second place prize of $35,000 in the annual Texas A&M New Ventures Competition. The event was held last Thursday, May 19th at Texas A&M University’s Memorial Student Center where TriFusion Devices competed against fifteen other student startups looking for help taking their idea to the next level. This was just the latest in a string of several startup competition wins for the team at TriFusion Devices, which is hoping to disrupt and revolutionize the healthcare, sports and protective equipment industries.
Last week TriFusion Devices, an up and coming 3D printing company launched by three Texas A&M students, added another startup competition win to their rapidly growing pot of seed money. The company took home the second place prize of $35,000 in the annual Texas A&M New Ventures Competition. The event was held last Thursday, May 19th at Texas A&M University’s Memorial Student Center where TriFusion Devices competed against fifteen other student startups looking for help taking their idea to the next level. This was just the latest in a string of several startup competition wins for the team at TriFusion Devices, which is hoping to disrupt and revolutionize the healthcare, sports and protective equipment industries.
Two million Americans are living with some form of limb loss, and an average of more than 500 limbs are lost each day. Despite those numbers, prosthetics are still exorbitantly expensive and even when they can be afforded, they often don’t fit as well as they should. Typical leg prostheses can cost more than $10,000 and they can take several weeks, even months to construct. When you consider that each prosthetic has a lifespan of about five years, it’s easy to understand why many amputees get frustrated with the entire system.
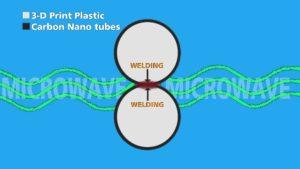
The TriFusion Devices team believes that they have come up with a system to make lightweight, high-quality, custom-fit leg prosthetic devices that can be delivered in 48 hours for a fraction of the cost of traditional devices. They developed a carbon nanotube-coated 3D printer filament that works in conjunction with a groundbreaking microwave welding process that will completely fuse together 3D printed parts. Not only does their process virtually eliminate the long wait to have a custom prosthetic manufactured, but it reduces materials and labor costs as well. The TriFusion materials technology even allows for post-printing fit adjustments of the final socket without slowing down the average two day manufacturing time.
Co-founders Blake Teipel and Brandon Sweeney, as well as Chief Medical Business Officer Britton Eastburn, pitched their new technology to a panel of judges that included venture capitalists, angel investors, entrepreneurs, and successful business executives. They competed against the other fifteen semi-finalists through three rounds of presentations, as well as a round that included an elevator pitch. The event is sponsored by the Texas A&M Engineering Experiment Station and the Texas A&M University System to recognize Texan startups with high-growth potential. The competition puts $100,000 of seed funding up for grabs to help the winning companies launch their ideas and start developing products.
Taking second place at the Texas A&M New Ventures Competition is the latest in a string of high profile student-centered business plan competitions. In 2015 the TriFusion Devices team won the 2016 Baylor New Venture Competition, Mays Business School’s Center for New Venture and Entrepreneurship Raymond Ideas Challenge, and the 2015 SEC Entrepreneurial Pitch Competition. Just last month they took home the first place prize from the prestigious Rice Business Plan Competition, taking home nearly $400,000 worth of startup funds and prizes. Discuss in the TriFusion 3D Printing Startup Second Prize Take Home forum over at 3DPB.com.
Take a look at TriFusion Devices elevator pitch:
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Unpeeled: New Arkema Material for HP, Saddle and Macro MEMS
A new Arkema material for MJF is said to reduce costs per part by up to 25% and have an 85% reusability ratio. HP 3D HR PA 12 S has been...
3D Printing News Briefs, January 20, 2024: FDM, LPBF, Underwater 3D Printer, Racing, & More
We’re starting off with a process certification in today’s 3D Printing News Briefs, and then moving on to research about solute trapping, laser powder bed fusion, and then moving on...
3D Printing Webinar and Event Roundup: December 3, 2023
We’ve got plenty of events and webinars coming up for you this week! Quickparts is having a Manufacturing Roadshow, America Makes is holding a Member Town Hall, Stratafest makes two...
Formnext 2023 Day Three: Slam Dunk
I’m high—high on trade show. I’ve met numerous new faces and reconnected with old friends, creating an absolutely wonderful atmosphere. The excitement is palpable over several emerging developments. The high...