Vanderbilt Researchers Spins Threads for Bioprinted Capillaries on Cotton Candy Machine
 While everyone gets pretty excited when researchers look to nature for new designs and innovation, taking a page out of the candy recipe book should certainly pique everyone’s attention, via that sweet tooth so many of us possess. Cotton candy evokes memories of the state fair, carefree afternoons, and delectable sugar crystals melting on our tongues, one gigantic artificially pink and blue colored bite after another. For researchers at Vanderbilt University though, it led them to a gigantic leap in science—as well as the revelation that making cotton candy is a lot more difficult than you may think.
While everyone gets pretty excited when researchers look to nature for new designs and innovation, taking a page out of the candy recipe book should certainly pique everyone’s attention, via that sweet tooth so many of us possess. Cotton candy evokes memories of the state fair, carefree afternoons, and delectable sugar crystals melting on our tongues, one gigantic artificially pink and blue colored bite after another. For researchers at Vanderbilt University though, it led them to a gigantic leap in science—as well as the revelation that making cotton candy is a lot more difficult than you may think.
By now, most everyone is aware of incredible advances being made via bioprinting, with the idea that it will one day lead to the creation of organs for transplants, thus changing—and saving—many lives in the process. We follow countless stories today regarding processes that are indeed allowing for the creation of cellular structures such as liver tissue, kidney tissue, and even amazing new tools to manipulate such bioprinted creations.
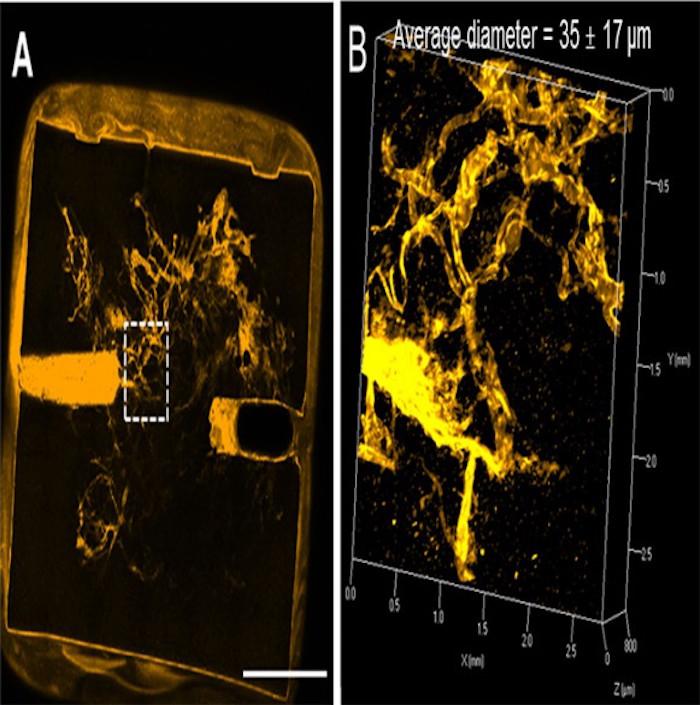
Microvascular network perfused with liquid. Figure B is magnification of the area in Figure A outlined in white. [Image: Bellan Lab / Vanderbilt]
What do we need to keep cells alive? Blood vessels. Capillaries, to be specific. And as if the challenge of learning to make cells in the lab through bioprinting wasn’t enough—indeed, now we must make capillaries to feed them. Bellan was inspired by the way that cotton candy machines spin threads of sugar to make the confectionary treat that’s been delighting humans for generations. Rejecting the methods used in bioprinting that support what he calls a ‘bottom up’ approach growing cells on a slab of gel, Bellan theorizes that his more refined ‘top down’ approach works much better in terms of speed and feasibility for this application.
“So far the other top-down approaches have only managed to create networks with microchannels larger than 100 microns, about ten times the size of capillaries,” explained Bellan.
He is able to create more complex networks through a new offshoot of the process of ‘electrospinning,’ harkening back to his days in graduate school where he actually created the exercise, making nanofibers from magnetic fields. For this very important new project—and theory—he definitely adjusted his original process, and the top-down process of his yields channels ranging from 3-55 microns with a mean diameter of 35 microns.
“The analogies everyone uses to describe electrospun fibers are that they look like silly string, or Cheese Whiz, or cotton candy,” offered Bellan. “So I decided to give the cotton candy machine a try. I went to Target and bought a cotton candy machine for about $40. It turned out that it formed threads that were about one tenth the diameter of a human hair–roughly the same size as capillaries–so they could be used to make channel structures in other materials.”
Indeed, this may be a way to create the networks which would nourish cells, and ultimately organs as they supply oxygen and remove waste. Previous methods using hydrogels, while serving as a foundation for this work, had to be refined.
“First, the material has to be insoluble in water when you make the mold so it doesn’t dissolve when you pour the gel. Then it must dissolve in water to create the microchannels because cells will only grow in aqueous environments,” Bellan added.

Three-dimensional slab of gelatin that contains a microvascular network. [Image: Bellan Lab /Vanderbilt]
Bellan used this material for the spinning of ‘threads’ in his cotton candy machine. A complex process is used to make a heated glue-like substance which is poured over the PNIPAM and forms an eventual gel for incubation—after which the cells and fibers are extracted. The fibers dissolve, leaving the network of what will eventually be blood vessels.
“Our experiments show that, after seven days, 90% of the cells in a scaffold with perfused microchannels remained alive and functional compared to only 60% to 70% in scaffolds that were not perfused or did not have microchannels,” Belan described.
“Our goal is to create a basic ‘toolbox’ that will allow other researchers to use this simple, low-cost approach to create the artificial vasculature needed to sustain artificial livers, kidneys, bone and other organs,” added Bellan.
Their next goal, upon this success, is to go another step further in getting more specific in terms of organs and bone as they begin to match specific tissues with the blood vessel networks, testing for true compatibility. One note here as well is the amount of progress these scientists have made using a $40 machine when the amount of funds put into machinery for labs is considered in comparison. What are your thoughts on this progress in bioprinting? Discuss in the Cotton Candy Helps with Bioprinting Capillaries forum over at 3DPB.com.
[Source: Forbes]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Srini Kaza Discusses Strategically Scaling Align’s “Smile-Changing” 3D Printed Aligners
Align Technology‘s Invisalign is a revolutionary method to get you the smile you want through 3D printing. It is also a hugely popular process to go through, a $4 billion...
Analysis: Additive Manufacturing Strategies Spotlights Vertical Value Creation
A slowdown in the industry and particularly a tightening of access to capital following the additive manufacturing (AM) industry’s peak special purpose acquisition company (SPAC) phase in early 2021 is...
Department of Defense Awards PROTECT3D $1.3M for Custom 3D Printed Braces
Custom prosthetics company PROTECT3D has secured a $1.3M Department of Defense (DoD) Small Business Innovation Research contract to develop a platform for creating custom braces for soldiers. The PROTECT3D Platform...
AMS 2025 Highlights Big Changes in 3D Printing with Pivotal Speakers and Panels
2023 was filled with excitement around the potential mergers being pursued by industry stalwart Stratasys (Nasdaq: SSYS), leading to some of the most insightful conversations imaginable at Additive Manufacturing Strategies...










































