Cyfuse Biomedical Partners with Cell Applications Inc. for the First Use of Regenova 3D Bioprinter Outside Japan
 When it comes to 3D bioprinting, there are a few companies who tend to dominate the news, while others labor more quietly in the background. Back in August, we wrote a feature on some of those lesser-known but significant companies in the bioprinting industry. One of those companies was Cyfuse Biomedical, a Japanese corporation that has developed a novel form of bioprinting known as the Kenzan method.
When it comes to 3D bioprinting, there are a few companies who tend to dominate the news, while others labor more quietly in the background. Back in August, we wrote a feature on some of those lesser-known but significant companies in the bioprinting industry. One of those companies was Cyfuse Biomedical, a Japanese corporation that has developed a novel form of bioprinting known as the Kenzan method.
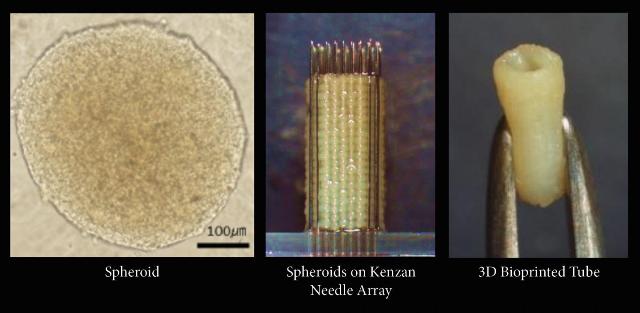
Most existing bioprinting techniques depend on scaffolding, which is solution made from biomaterials such as collagen or hydrogel. The solution is required to hold together the seed cells that will be grown into tissues or organs. With the Kenzan method, no scaffolding is needed. “Kenzan” is Japanese for “needle array,” and an array of needles form the basis of the process. Cyfuse’s Regenova bioprinter works by gathering cell aggregates, or spheroids, onto a series of needles in a pattern predetermined by 3D software. The needles are so close together that the cells they hold will fuse together into solid, firm tissue when placed in a bioreactor.
Cyfuse began marketing the Regenova printer not long ago, and now a new partnership with San Diego-based Cell Applications, Inc. will make tissue produced by the Kenzan method available to North American researchers – the first time the Regenova printer will be used outside of Japan.
“The Regenova 3D Bio Printer, combined with Cell Applications’ comprehensive, high-quality primary cell bank, offers researchers streamlined access to a nearly limitless selection of three dimensional tissues including those mimicking blood vessels, human neural tissue and liver constructs,” said Koji Kuchiishi, CEO of Cyfuse Biomedical. “The collective strengths of both our companies will serve the growing demand for viable engineered tissues and accelerate scientific discovery in North America, taking us one step closer to making regenerative medicine a reality.”
 Cell Applications will utilize the Regenova printer to produce tissue for researchers on a pay-for-service basis. The company already offers over 100 types of animal and human primary cells in about 900 configurations, and their custom services program isolates certain types of primary cells upon request. With the Kenzan method, more delicate primary cells can be preserved than in other bioprinting methods, in which cells are frequently damaged. The Kenzan method yields a higher amount of viable cells that maintain the critical properties of their original tissue systems, without any kind of chemical or genetic modification.
Cell Applications will utilize the Regenova printer to produce tissue for researchers on a pay-for-service basis. The company already offers over 100 types of animal and human primary cells in about 900 configurations, and their custom services program isolates certain types of primary cells upon request. With the Kenzan method, more delicate primary cells can be preserved than in other bioprinting methods, in which cells are frequently damaged. The Kenzan method yields a higher amount of viable cells that maintain the critical properties of their original tissue systems, without any kind of chemical or genetic modification.
“In addition to customized cell isolation and assay services, Cell Applications is now able to provide researchers with an integrated cell-engineering solution that utilizes our expansive primary cell bank and the innovative Kenzan bioprinting method,” said James Yu, founder and CEO of Cell Applications. “Having the Regenova 3D Bio Printer at our San Diego headquarters with our vast array of primary cells is a powerful combination. We’re very pleased to offer researchers an end-to-end, customized solution for creating scaffold-free, 3D-engineered tissues that reduce costs by minimizing the lengthy processes typical in pharmaceutical drug discovery.”
So far, academic research institutions have been using the Kenzan method to print nerves, blood vessels, and liver-like tissue. The potential future applications of the technique are numerous – pancreatic islets, cardiac muscles, and production of skin, spinal cord, and other types of tissues for drug screening. And, like all other bioprinting organizations, Cyfuse hopes that their technology will eventually lead to the printing and regeneration of entire, functional human organs. Discuss this latest news in the Cyfuse 3D Bioprinter forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Making 3D Printing Personal: How Faraz Faruqi Is Rethinking Digital Design at MIT CSAIL
What if your 3D printer could think more like an intelligent assistant, able to reason through a design idea, ask questions, and deliver something that works exactly the way the...
Reinventing Reindustrialization: Why NAVWAR Project Manager Spencer Koroly Invented a Made-in-America 3D Printer
It has become virtually impossible to regularly follow additive manufacturing (AM) industry news and not stumble across the term “defense industrial base” (DIB), a concept encompassing all the many diverse...
Heating Up: 3D Systems’ Scott Green Discusses 3D Printing’s Potential in the Data Center Industry
The relentless rise of NVIDIA, the steadily increasing pledges of major private and public investments in national infrastructure projects around the world, and the general cultural obsession with AI have...
Formlabs Teams Up with DMG MORI in Japan
In late June, Nick Graham, Chief Revenue Officer at Formlabs, announced on LinkedIn that the company had partnered with DMG MORI, one of the world’s leading machine tool companies, to...