Swansea University Scientists Engineer 3D Bioprinted Tissues for Pharmaceutical Research
It has been announced, that a novel application of 3D-Bioprinting living tissue models for pharmaceutical testing is being developed at Swansea University-UK. A team of scientists have begun trailing a process, which has been designed to augment the development of new drugs and treatments. The research team led by Dr. Daniel Thomas of the Welsh Centre for Printing and Coating has produced a process which uses precision deposition technologies to produce complex tissue architectures.
Over the past decade, annual spending on pharmaceutical development has doubled to more than £30 billion per annum. At the same time, in spite of these huge sums invested, the average number of drugs being approved for human use has decreased to one in ten. Currently, pre-clinical studies to test the safety and efficiency of new drugs use laboratory animals and traditional 2D cell culture models. Neither of these methods are completely accurate reflections of how a drug will react in a human.
A solution has emerged in the form of 3D-Bioprinting technology developed for the scalable, accurate and repeatable deposition of biologically active materials. With advances in this bio-manufacturing technology, durable biological tissues for use in testing new pharmaceutical products are now being harnessed and refined.
Dr. Thomas explains “Using self-differentiation 3D bioprinting processes, then it is not necessary to print all of the details required to form a tissue. The concept is that once the relevant cells are placed in the right place, nature completes the process. This is as such a top down method for engineering new ways towards making tissue structures, which is no different to cells in an embryo knowing how to configure themselves into complicated organs. Nature has been evolving this capability for four billion years, and we can use this process to our advantage. Thus we do not have to print networks of vasculature and innervated layers using this approach, as these can be produced by using specific three dimensional stem cell differentiation processes.”
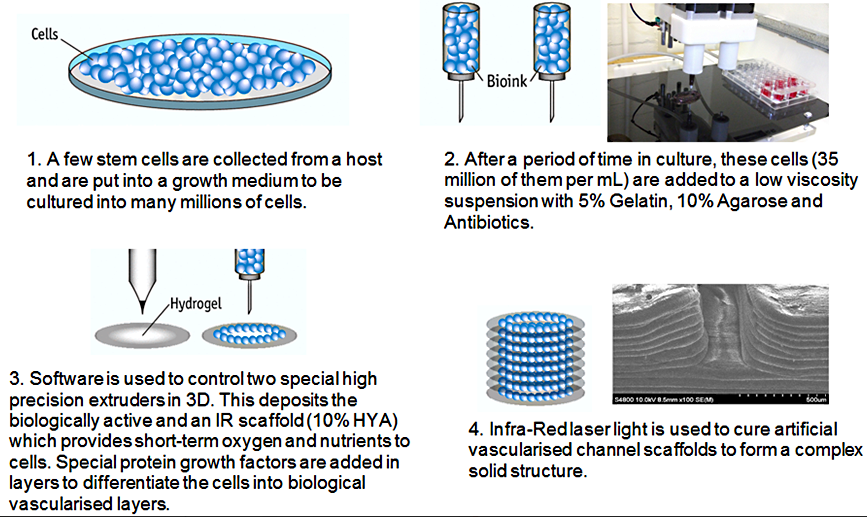
Overview of the 3D-Bioprinting process, used to produce experimental coupon tissues for pharmaceutical testing.
Going forward, 3D Bioprinting is being explored as a method for the creation of more advanced structures. In the longer-term, this technology offers the potential to fabricate organised tissue constructs. This is being engineered to repair and/or replace damaged or diseased human tissues, and directly has a bearing for developing safer and more effective healthcare treatments. It also opens up the opportunity for cost effective patient specific tissue engineering to evolve. However, fundamental obstacles include balancing scaffold properties to optimise; resolution, cell migration, proliferation and differentiation need to be overcome, one step at a time.
“By further engineering this process then we can produce tissues which have measurable mechanical, metabolic and functional properties. This is from the perspective of using shaped scaffold bioprinting technology which produces complex structures.” said Dr. Thomas.
This process specifically deposits an Extra Cellular Matrix (ECM) protein scaffold in 3D. ECM is a biochemical support material between all cells and is critical towards the formation of a tissue structure.

Extra-cellular matrix 3D Bioprinting, which is a method engineered to generate protein scaffolds. These are later transferred into an incubator which contains stem cells and transforming growth factor alpha.
The potential to produce functional tissues on demand, made in a controlled and safe way for use in humans could one day revolutionize healthcare. In the future, 3D-Bioprinting technology could have a huge global impact on healthcare and the economy.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: 2024 Results In, Nano Dimension Gears Up with Markforged and Desktop Metal
Nano Dimension (Nasdaq: NNDM) is going through a major transformation. In just a few weeks, the company has finalized not one, but two major acquisitions: Desktop Metal and Markforged. On...
Velo3D Secures $22M Metal 3D Printing Powder Supply Deal with Amaero
Velo3D (OTCMKTS: VLDX) has signed a major new agreement with metal 3D printing powder manufacturer Amaero (ASX: 3DA). The five-year deal is valued at USD $22 million and centers around...
3D Printed Heat Exchanger Specialist Conflux Technology Expands into the UK
Melbourne’s Conflux Technology, a specialist in using additive manufacturing (AM) to produce heat exchangers, is opening a business hub in the UK. The new site, which will open sometime in...
Rob Higby: How Continuum’s Scrap-to-Powder Model Caught Siemens’ Attention
Continuum Powders has spent over a decade working on a bold idea: why mine new metal when high-quality material is already flowing through today’s factories as scrap? Now, thanks to...






























