Bioprinter Maker Rokit Prepares for Korean IPO
Leading bioprinter manufacturer Rokit Healthcare is gearing up for its Initial Public Offering (IPO) as early as July 2021. In March 2021, the South Korean company announced it is preparing a listing on the Korean Stock Exchange and will issue 12 million shares. Organized by Seoul financial service providers KB Securities and Mirae Asset Financial Group, the move could strengthen Rokit’s overseas strategy and support its 4D bioprinting innovations. This is true particularly now that the company has already begun commercializing its flagship bioprinted skin patches to treat diabetic foot ulcers and started clinical trials of a platform to help regenerate damaged cartilage tissue in patients with arthritis ulcers.
“We are currently in the process of two technology evaluations and are anticipating authorization results for the IPO in mid-March 2021,” said Rokit Founder and CEO Seok Hwan You to news outlet BioWorld. “Focused on personalized therapy solutions, You even said that “one of our American investors has a father who suffers from diabetic foot ulcers, and this was a big reason why he decided to invest in our company.”
South Korea’s healthcare market is ranked fourth among countries in the Asia-Pacific region due to a combination of increased insurance coverage, an aging population, and government initiatives. In November 2020, South Korea released a new law on “Advanced Regenerative Medicine and Advanced Biopharmaceuticals.” Debated for years, the legislation is designed to better align Korea with similar regulations in force in Western Europe and the United States by creating product definitions and establishing parameters for their clinical research, monitoring, and surveillance.
Moreover, South Korea’s plans to invest more than $1.7 billion in its biotechnology and biopharmaceutical sectors over the next five years is slated to attract foreign investment. This growing healthcare market has led to the evolution of several niche biotech companies, like Rokit. In fact, according to a report recently released by the Korea Exchange, IPO’s of companies in biopharma is booming, replacing manufacturing and finance firms. In July 2020, shares of the biotechnology group SK Biopharmaceuticals soared on its stock market debut, in what was South Korea’s biggest IPO in three years. By year-end, Seoul’s market saw a record-breaking streak of bio IPOs.
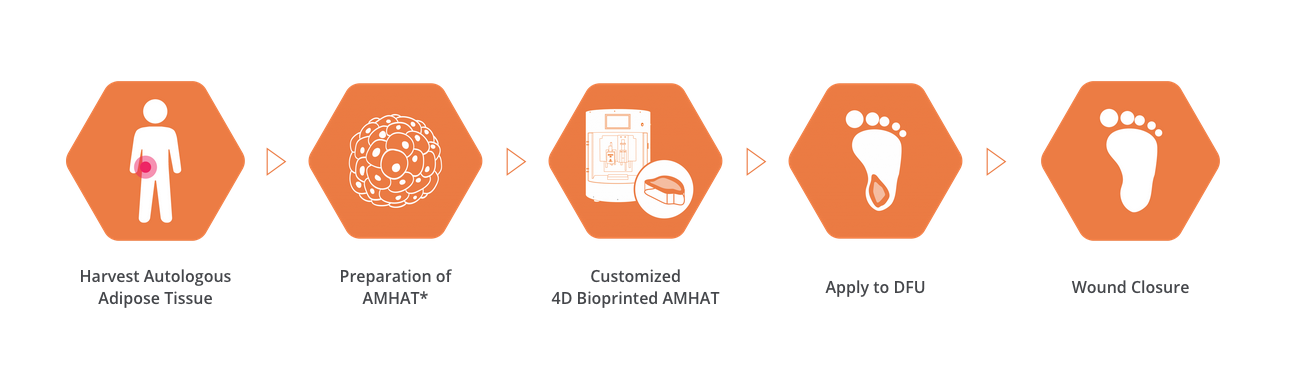
This is a very promising scenario for Rokit and its regenerative medicine aspirations. The business has made headway with many of its innovative products in the past two years. Among its customized artificial organ technologies, Rokit’s diabetic foot ulcers Regeneration Platform (DFURegen) is a practical, personalized regenerative therapy for treating diabetic foot ulcers (DFU). The one-time treatment doesn’t require hospitalization and is derived from the patient’s fat tissue, which is used to prepare an autologous extracellular matrix to form a bioink. The material is then loaded into Rokit’s Dr. INVIVO DFU 4D bioprinting platform to produce a dermal patch that is then applied to the DFU where the wound closes.
The platform was successfully tested on patients in Korea and India, with results from these studies showing 100% wound healing for all tested patients with chronic DFUs. According to data published by Rokit in the American Diabetes Association‘s journal Diabetes, the autologous extracellular matrix patch applied onto the chronic wound site of DFU patients resulted in a significant wound size reduction after only a one-time treatment. During the tests, most of the patients showed complete wound closure in just two to five weeks. In fact, after a 14-day skin wound healing process, the treated wound area had more effectively reconstructed epidermal and dermal structures when compared to the non-treated wound areas.
In 2020, the innovative product began commercializing in 48 countries, including India, countries in Europe, Latin America, the Middle East, and North Africa. However, the company already has plans to expand, launching the platform in other regions, like North America, Japan, China, and Australia. Rokit has two ongoing clinical studies at the LewisGale Medical Center in Virginia and Ankara City Hospital in Turkey to take their product to these new markets.
Rokit is also using its INVIVO 4D bioprinting technology to treat osteoarthritis in the first human case of cartilage regeneration treatment. The company has already collected promising efficacy results from the collaborative pre-clinical trial project with Massachusetts General Hospital in Boston and is currently verifying and validating the procedure on a clinical scale at Assiut University Hospitals in Egypt.
Similarly, on November 23, 2020, Rokit began a sizable preclinical animal model trial for HumaTein Skin Spray, a product specially developed for treating soldiers with traumatic burn injuries in the field. HumaTein Skin Spray uses HumaTein technology that prevents infection as a first-aid and induces simultaneous skin regeneration that could dramatically reduce scar formation and hospitalization length to enhance patients’ quality of life. Rokit could also use the 4D technology to regenerate entire organs, with the company successfully creating kidney cells in recent animal trials.
Founded in 2012, Rokit is one of the fastest-growing bioprinting companies globally, with 110 employees working in six business units. Its cutting-edge organ regeneration platforms based on 4D printing and clinical diagnosis technology for age-related diseases, such as rheumatoid arthritis and Alzheimer’s disease, will be crucial to the anti-aging market worldwide.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Scientists Create Stretchy 3D Printed Implants for High Blood Pressure Treatment
Researchers at Pennsylvania State University (Penn State) say they may have found a softer, less invasive way to treat severe high blood pressure. In a new study published in the...
Harvard’s Jennifer Lewis Lab Is 3D Printing Artificial Muscles That Twist and Bend on Demand
Researchers at Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) have developed a new way to 3D print materials that can move on their own, bending, twisting,...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Harvard SEAS Engineers Develop 3D Printing Method for Soft Robotic Components with Programmable Shapes
The world of soft robotics is still largely in its pure research phase, but the R&D landscape has started to produce examples of early-stage commercialization. Researchers have started to refine...