Many companies have been working on alternatives to needle-based drug and vaccine delivery as well as for minimally invasive patient testing. One extensively investigated alternative in recent decades has been microneedle arrays, a type of minimally-invasive device that painlessly bypasses the skin’s principal barrier to topically-apply drugs.
But this approach is not just about needle-free drug delivery. In order to fight disease, there is a strong need to engineer systems for vaccination through the skin, the most accessible human tissue due to its sensitivity and very reactive immune response. Recently, researchers from the University of Pittsburgh proposed the development of a novel device for cutaneous vaccination based on microneedle arrays (MNA) fabricated from water-soluble polymers, using a two-photon polymerization 3D printer from Nanoscribe, a high-precision additive manufacturing pioneer.
Currently, most vaccines use painful hypodermic needle-based injections that deliver antigens into the muscle or subcutaneous tissues, which contain fewer immune cells than viable skin does. Moreover, administering these traditional vaccines requires trained healthcare personnel, excessive costs from cold chain storage and transport, and risks of disease transmission and needle-stick injuries. To tackle this issue, the interdisciplinary team of scientists and engineers came up with dissolving MNAs for skin vaccine delivery systems.

Dissolving MNAs with materials loaded on the pyramid region of the microneedles (Credits: University of Pittsburgh)
In a paper published in the Journal of Controlled Release, the researchers described the first study introducing an additive manufacturing strategy to fabricate fully dissolving skin-targeted MNAs, thereby eliminating complicated engineering procedures.
As stated in the study, MNAs are composed of micron-scale needles with sharp pyramid-shaped heads supported by undercut stem regions with filleted bases to ensure successful skin penetration and retention during application. The MNAs are fabricated from water-soluble polymers that, in their dry-state, are strong enough to penetrate the outermost layer of the skin, and then rapidly dissolve and release their cargo upon insertion into the hydrated skin tissue. The project ultimately demonstrates the capacity of dissolving MNAs to precisely deliver vaccines to cutaneous microenvironments with improved efficiency, requiring relatively lower vaccine doses compared to traditional injections.
Researchers used a Nanoscribe Photonic Professional 3D printing system at the university’s Nanoscale Fabrication and Characterization Facility to print prototypes and masters of MNAs that enabled the fabrication of dissolving microneedle arrays, which are expected to facilitate diverse immunization strategies.
The 3D microfabrication of microneedle array prototypes involves six steps, beginning with a CAD design of the targeted MNA, which is used to produce the master MNA by 3D direct laser printing. Followed by a high-fidelity replication of the master MNA using a two-step micro-molding approach, and then the creation of the MNA master molds that consist of multiple master MNA replicas on 3D-printed MNA holders. This is succeeded by the manufacturing of MNA production molds that are used to finally fabricate tip-loaded, dissolving undercut MNAs incorporating the target vaccine.
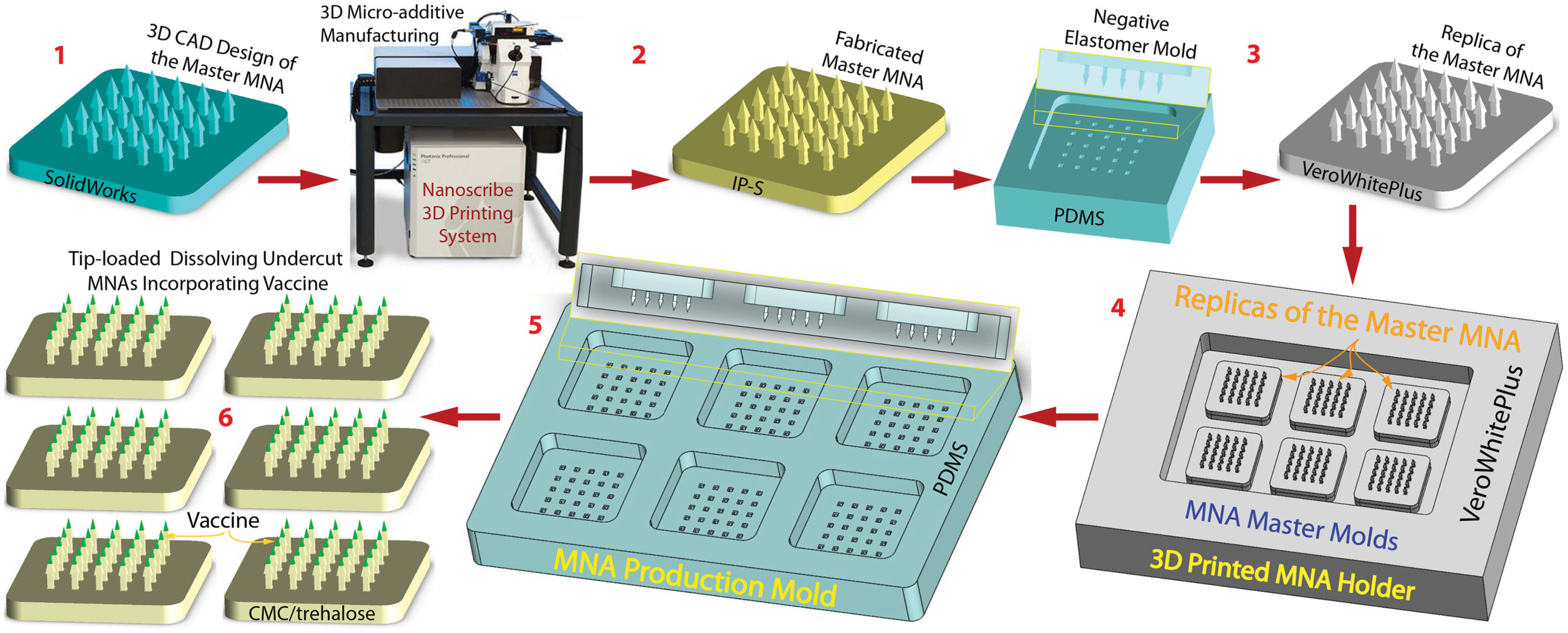
Microneedle array manufacturing strategy involves six distinct steps (Credit: University of Pittsburgh)
While traditional 3D printing methods like stereolithography and fused deposition modeling have been used to produce non-dissolvable microneedle arrays, the authors considered that those technologies were not enough to achieve the nanoscale resolution and design capabilities provided by Nanoscribe’s technology.
Furthermore, Nanoscribe claimed that when the researchers were “designing microneedle arrays for cutaneous drug or vaccine delivery, Nanoscribe’s 3D printer allowed them to think freely without worrying about the design complexities, while also offering great ease of use.” They also reported that “Nanoscribe’s systems not only attract engineers, but also impress medical scientists who do not necessarily have microfabrication expertise, and look for simple ways to realize their ideas. For both disciplines, 3D microfabrication has become a key technology to achieve the optimization of cutaneous drug or vaccine delivery systems.”
Ultimately, the team successfully tested its MNA in vivo. The 3D-printed MNAs with antigens were applied to abdominal skin in mice for periods of between five to 20 minutes to prove the efficient delivery of the multi-component vaccine. More importantly, they claim to have proved that skin vaccination with antigen-loaded MNAs elicited more potent antigen-specific cellular and humoral immune responses than traditional immunization by intramuscular injection.
Novel drug delivery systems, such as microneedle arrays, can benefit from replication processes needed to upscale production. These advances will help move toward the scalability required in industrial manufacturing for the widespread application of skin vaccination.
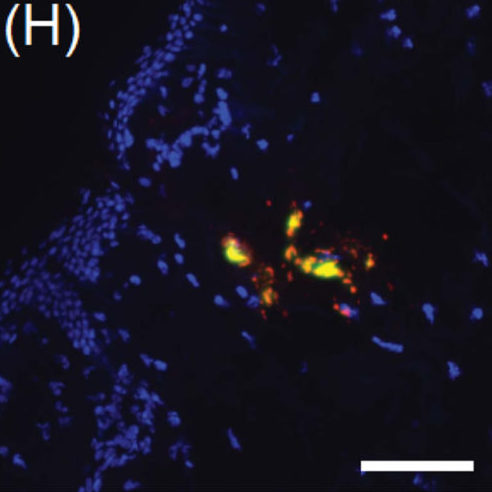
Intradermal vaccine delivery to freshly-excised human skin explants using tip-loaded dissolving MNAs with undercut microneedles (Credit: University of Pittsburgh)
This novel form of drug delivery is being developed at a time when researchers around the world are racing to develop vaccines to safely and efficiently prevent the spread of a variety of diseases, particularly COVID-19, caused by the SARS-CoV-2 coronavirus. In fact, Louis Falo, a co-author of the paper and researcher at the University of Pittsburgh’s School of Medicine, also recently announced that along with colleagues, they have developed a potential vaccine against SARS-CoV-2 that has been successfully tested in mice, and was administered using MNA patches that delivered biocargo to the skin to increase potency.
With the potential to impact a broad range of cutaneous and non-cutaneous vaccination and drug delivery applications, the new vaccine delivery system can be a critical component of the future of immunization delivery methods. Introducing antigens to the body through MNA patches rather than needles can change the way many people access vaccines, especially in developing countries, rural areas, and impoverished regions, where there are limited health providers qualified to administer injections and the conditions for current vaccine transportation are costly and hard to come by. Every year, nearly 1 in 10 children do not receive any vaccinations, and more than 1.5 million people worldwide die from vaccine-preventable diseases. If vaccine research and testing can change application methods, scientists will broaden the availability and facilitate access to immunization that can help prevent untimely deaths.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
ExOne + voxeljet Are Trying to Do the One Thing Customers Need Right Now: Keep Machines Running, and Rebuild Confidence
For years, ExOne and voxeljet were two of the best-known names in binder jet 3D printing, especially in sand printing for foundries. But in recent years, ExOne “got buried” inside...
BigRep Launches ONE.5X 3D Printer, Announces New Massive Dimension Partnership at RAPID + TCT 2026
As the whole world is starting to realize, the Hormuz supply chain fallout is only just beginning to filter into the global economy, and the rising cost of plastics should...
Orano Federal Services & UNC Charlotte Show How AM Could Cut Costs in Nuclear Energy Resurgence
Outside of the defense sector, few industries have been impacted by Russia’s ongoing occupation of Ukraine more than nuclear energy. The same appears to already be happening in response to...
EOS to Spotlight AI, Robotics, and Industrial Tooling at Hannover Messe
The US-Israel war on Iran is already catalyzing the sorts of major shifts to global supply chains that will effectively amount to permanent economic changes. In this context, the nations...































