US Researchers Provide Holistic Perspective on Extrusion-Based 3D Printing of Personalized Medicine
US researchers continue ongoing research into 3D printed pharmaceuticals, presenting their unique findings in ‘Polymers for Extrusion-Based 3D Printing of Pharmaceuticals: A Holistic Materials-Process Perspective.’
While many have predicted that the pharmaceutical industry was perched on the edge of a massive transformation due to the advantages offered by 3D printing, there are still many ongoing studies regarding suitable printers, materials, and critical issues such as the impacts of DIY drugs and more formalized manufacturing. In this study, the researchers examine both extrusion-based FDM 3D printing and pressure-assisted micro-syringe 3D printing as they compare them to more traditional processes, along with techniques, challenges, and consider the general direction of the industry overall.
Within the pharmaceutical industry today, 3D printing has made impacts in digital fabrication of drugs, implants, drug delivery systems, and more; however, the researchers point out the importance of such processes especially in creating personalized medications. Citing the success, and FDA approval, of epilepsy drug SPRITAM®, the authors stress advantages in the trend toward patient-specific treatment—and specifically, medicine.
“Current pharmaceutical manufacturing practices are not cost‐effective for personalized medicine,” state the researchers. “3D printing pharmaceuticals are more suitable than current manufacturing practices for tailored solid dosages.”
Although the ‘first waves’ of pharmaceutical 3D printing began with inkjet printing, strides since then have been not only impressive, but fascinating to much of the public, including:
- Powder-based printing
- Extrusion
- Pressure-assisted microsyringes (PAM)
- Stereolithographic printing (SLA)
- Selective laser sintering (SLS)
- Inkjet printing
- Digital light processing
FDM and PAM printing processes have been most attractive to users due to accessibility and affordability, the potential for fabricating and tuning complex geometries, and more.
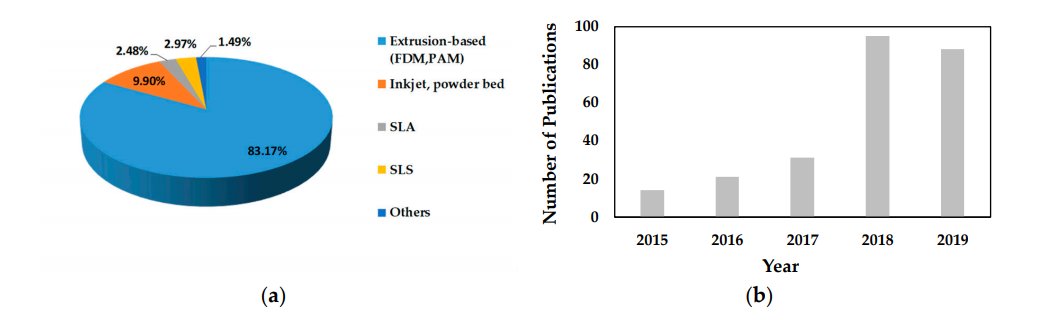
(a) The proportion of research articles published on different types of 3D printing processes in the last five years (2015‐2019, total 202 articles); (b) The number of published scientific articles (research and review) in the period from 2015 to 2019 which reported the use of extrusion‐based (fused deposition modeling (FDM) or pressure‐assisted microsyringe (PAM)) 3D printing (source: Scopus database and PubMed).
Choosing the right materials is critical to the success of 3D printing medications also, along with the capability for attaining FDA approval. As a highly suitable material, polymers may be used to print the following in relation to tablets and caplets:
- Binders
- Disintegrants
- Compression aids
- Diluents
- Fillers
- Drug release
“Polymers’ multifaceted utilization in solid dosage drug delivery systems solidifies its importance in pharmaceutical 3D printing applications,” state the researchers.
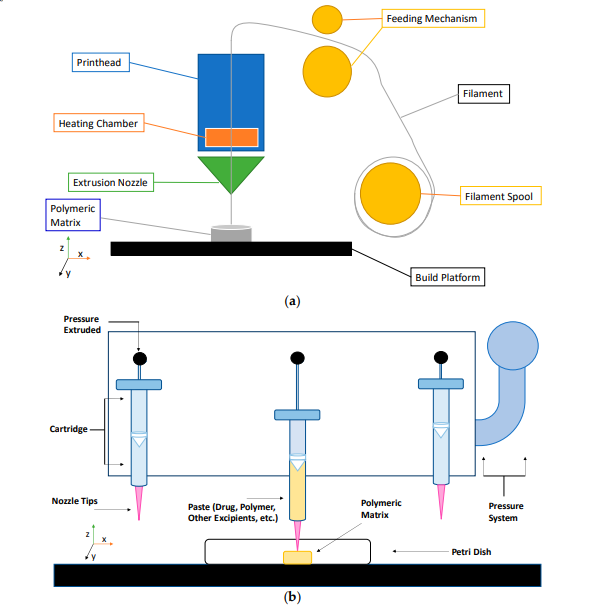
2‐dimensional (2D) schematic of the extrusion‐based 3D printing process: (a) fused
deposition modeling (FDM), (b) pressure‐assisted microsyringe (PAM).
And while they have reviewed studies from multiple other scientists, the authors here point out that while such guides are needed, no ‘detailed discussion’ on how polymers should be selected—along with other details—exists.
Processing is very similar via FDM or PAM, with the extra benefit of being more environmentally friendly, offering the ability for remote control of printing, and small batch, on-demand printing.

Comparison of different processing steps required for traditional direct compression (DC) tablet manufacturing vs. advanced manufacturing, 3D printing (FDM or PAM).
Typical polymers that can be used for 3D printing in pharmaceuticals include:
- Carbopol®
- Ethylcellulose (EC)
- Eudragit®
- Hydroxypropyl Cellulose (HPC)
- Hydroxypropyl Methylcellulose (HPMC)
- Polycaprolactone (PCL)
- Polylactic Acid (PLA)
- Polyvinyl Alcohol (PVA)
- Polyvinylpyrrolidone (PVP)
- Poly(Ethylene Glycol) (PEG)
- Soluplus®

Summary of polymers based on (a) water solubility and drug release type, (b) their selection for either FDM or PAM 3D printing method.
Obviously, the pharmaceutical industry is highly regulated, with good reason, and the production of medications can be complex due to stringent requirements in production for the safety of the patient. In terms of 3D printing, however, a list of challenges still present themselves such as lack of predictability and consistency in quality of product due to issues like shrinkage or warping, problems with mechanical stability and other properties, and more.
“Though a large-scale production of pharmaceuticals using 3D printing might be a long way from now, personalized medicine is possible in‐house for immediate use,” conclude the researchers. “Future work to enable drug product manufacture using FDM and PAM 3D printing technologies should include the suitability and characterization of polymers and other excipients amenable to processing.
“Polymer materials and their properties, specifically their rheology should be investigated to allow a wider formulation and 3D printing design space. A better understanding of the rheological properties of API–polymer mixtures and their measurement is necessary for the successful 3D printing of pharmaceuticals.”
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.

Characterizations required for 3D printed structure, drug, and polymer (or other functional excipients, if necessary).
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Flashforge Bets on Meshy AI as Desktop 3D Printing Battle Intensifies
Competition in desktop 3D printing is brutal. Whereas before, firms competed through value engineering, Prusa clones now have an integrated hardware, sensor, and software setup that is making all the...
Asia AM Watch: Advantages to the Chinese Way of Doing Business
Timo Göbel, the Head of Additive Manufacturing at the BMW Group, spoke at the AM Forum in Berlin about industrializing additive. He had wise words to share, including that we...
3DPOD 300: Celebrating 300 Episodes with a Look at the Next Year in 3D Printing
In the 300th episode of the 3DPOD, we take a look at what we think will happen over the next 12 months; for instance, what will happen with Bambu’s dominance,...
3D Printing Financials: 3D Systems Returns to Growth in Q1 2026
3D Systems (NYSE: DDD) reported one of its strongest quarters in recent years, showing signs that the company may finally be moving past the tough slowdown that has weighed on...
















































