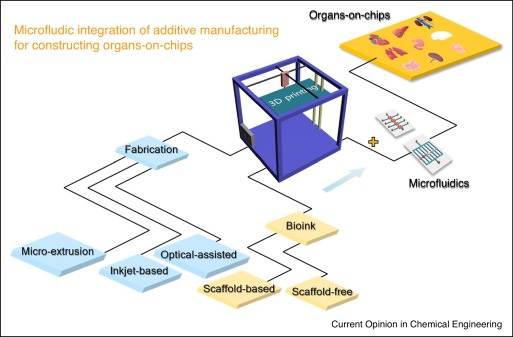
3D Printing, Microfluidics & Organ-on-Chips Coming Together
In the recently published ‘Combining additive manufacturing with microfluidics: an emerging method for developing novel organs-on-chips,’ Chinese researchers are exploring a complex but increasingly popular topic in 3D printing, combining the technology with devices like organs-on-chips (OOCs).
As additive manufacturing continues to spur on new developments in research and other areas such as education (in nearly every grade—all the way up to the highest graduate degrees) and engineering, greater innovation continues in OOCs, microfluidic platforms used to imitate the functionality of human organs.
While OOCs were initially much more rudimentary and lacking in necessary adjustability, today they are incredibly advanced as scientists move closer and closer to their goal of being able to transplant 3D printed organs into the human body with success. And while bioprinting has progressed immensely, the technique is still laden with challenges due to the delicate nature of tissue engineering.
Recently, new efforts have been made to bioprint with OOCs, including projects such as:
- Large-scale microfluidics
- Precise 3D cellular architectures
- Flow control for stable microenvironment maintenance
- Generation of tissue/organ-level structures
- Tissue-to-tissue interfaces

Schematics of 3D-cell printing methods with different working principles: (a1) micro-extrusion, (a2) inkjet-based and (a3) laser-assisted printing. Micro-extrusion based printing [12]. (b1) Rendering of the assembled syringe pump extruder and a printer. (b2) Time-lapse sequence of 3D bioprinting of a university logo. (b3) Printed collagen heart and the cross-sectional view of the collagen heart. Inkjet-based printing [16]. (c1) Schematic representation of 3D checkerboard composed of two patterns. Patterns of a university logo (c2), concentric circles, partial circles pattern and ‘Smiley face’ (c3) obtained by printing Fluo-ink (green) and Acri-ink (blue) containing Tomato NIH 3T3 fibroblasts (red). (g and h: scale bar 200 μm). (c4) A bioprinting hydrogel-based microfuidic chip. Scale bar: 1 mm. Laser-assisted printing [17]. (d1) Schematic of laser direct-write. (d2) Fabricated microbeads and laden cells. Scale bar: 200 μm. (d3) Confocal microscopy images of MDA-MB-231 3D aggregate. Scale bar: 100 μm. Reproduced with permissions from the American Association for the Advancement of Science [12] and Elsevier [17].
- Interact with cells
- Provide vehicles for cell loading
- Build scaffolds for tissue formation
They are often either naturally gleaned from materials like gelatin or alginate, as well as synthetics like polyethylene glycol and Pluronic©.
“In cell-laden hydrogels, biologically active components including growth factors, other extracellular matrix (ECM)-associated proteins are usually encapsulated for enhancing cell adhesion, cell proliferation or differentiation,” state the researchers. “Solidification of printed hydrogels is realized through thermal, photo cross-linking, or ionic/chemical cross-linking processes. Recently, hydrogel bioinks have been doped with nanomaterials for improving robustness and cell differentiation.”
As bioprinting continues to advance, we have seen:
- Liver-on-a-chip
- Bioprinted-heart-on-a-chip
- Vasculature-on-a-chip
Characterization continues in 3D bioprinted OOCs also, assessing both development and functionality using biochemical and biomechanical analyses. As the research team points out though, cell viability is an ‘essential parameter’ when it comes to OOC development. Biochemical studies are used to test OOCs with genetic and protein expression information also.
“In brief, from a view of printing resolution, the extrusion-based printing, which has been the most widely accepted is still not yet compatible for all design when the on-chip structures become more sophisticated and heterogeneous. SLA has a higher resolution, but the cell viability is inevitably affected during laser or UV light exposing,” conclude the researchers.
“In parallel, integration of embedded physical, biochemical and optical sensors with OOCs can record real-time cell behavior and environmental parameters. All these innovations will extend the applications of bioprinting integrated OOCs in fundamental research and clinical settings.”
Organ-on-a-chip technology continues to progress in labs around the world, from advanced engineering methods to prototypes to help cut costs and even the use of such methods to combat viral threats.
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
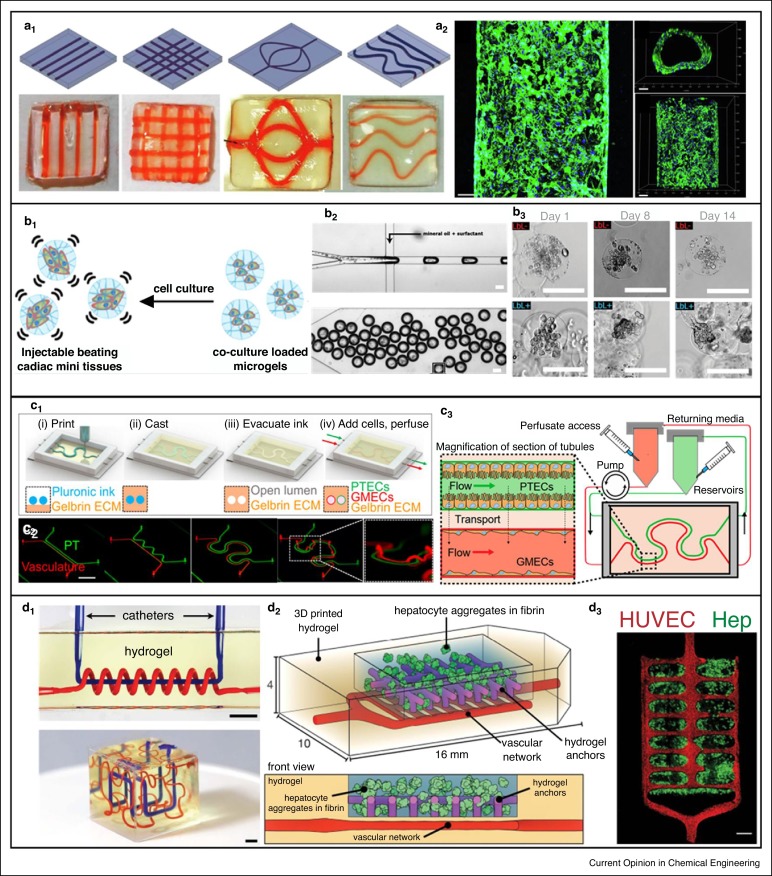
3D bioprinting integrated with microfluidic OOCs. Liver-on-a-chip [33]. (a1) Digital designs and corresponding devices (a2) Fluorescent images of the hUVECs cultured within channels (green showing F-actin and blue showing nuclei). Scale bar: 100 μm. Heart-on-a-chip [36]. (b1) Principle of on-chip functionalized microtissue by coculture of hiPSC-CMs and normal human cardiac fibroblasts. (b2) Microfluidic chip for cell-laden droplet generation (above) followed by transforming to microgels (below). Scale bar: 100 μm. (b3) Micrographs of cultured hiPSC-CM/NHCF-Vs at days 1, 8, and 14. Kidney-on-a-chip [39]. (c1) Schematic of 3D vascularized proximal tubule fabrication process. (c2) Fabricated on-chip vascularized proximal tubule. Scale bar: 10 mm. (c3) Integration of 3D vascularized proximal tubule tissue with a closed-loop perfusion for measuring renal reabsorption. Vasculature-on-a-chip [42••]. (d1) Adaptations of mathematical space-filling curves to entangled vessel topologies of axial vessel and helix (up) and interpenetrating Hilbert curves (down) within hydrogels. Scale bar: 3 mm. (d2) Hepatic hydrogel carriers created by seeding endothelial cells (HUVECs) in the vascular network after printing. (d3) Confocal microscopy images of hepatocyte aggregates (Hep) in fibrin gel entrapped by hydrogel anchors. Scale bar: 1 mm. Reproduced with permissions from Elsevier [33], the American Chemical Society [36], the National Academy of Sciences of the United States of America [39] and the American Association for the Advancement of Science [42••].
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
RAPID 2026 in Pictures: The Coolest & Craziest Things on the Show Floor
Last week, North America’s premier 3D printing trade show, RAPID+TCT 2026, came to Boston. I spent two days trekking the show floor, trying to see as much as I could,...
ExOne Cuts Costs for U.S. Customers as Printhead Production Moves to Detroit
ExOne Global Holdings, created through the 2025 integration of ExOne and voxeljet, is making changes across its U.S. operations. These include starting printhead manufacturing in the Detroit area and lowering...
Euler Viewer for Metal LPBF 3D Printing Released
Icelandic software startup Euler has released Euler Viewer, a real time build viewer for metal LPBF. The product does not need to be installed, and doesn’t require hardware to be...
3D Printing News Briefs, April 11, 2026: Energy Targets, DoW Contracts, Nike Air Max, & More
We’re starting with 3D printing for energy applications in this weekend’s 3D Printing News Briefs, and then moving on to military and defense 3D printing. Finally, Nike Sportswear is focusing...