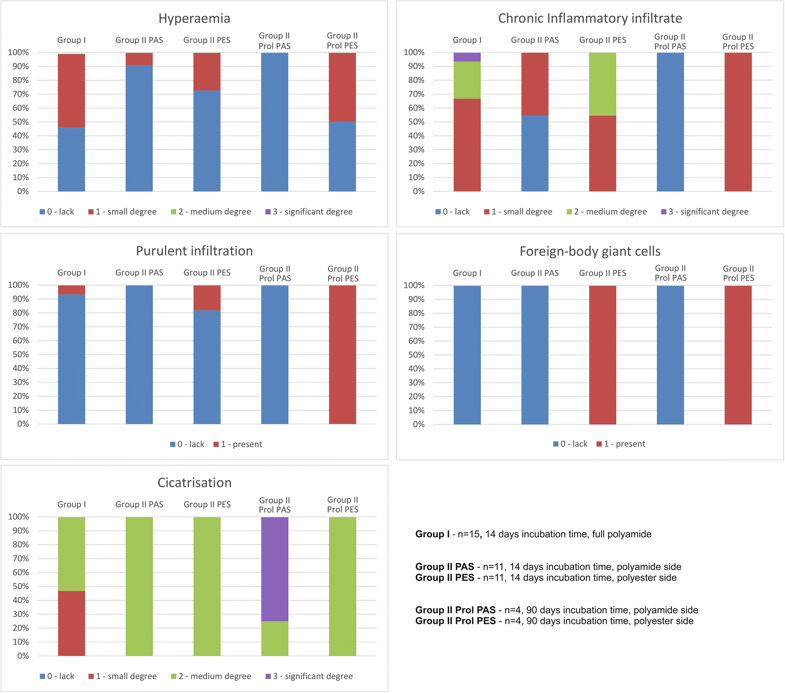
In ‘Evaluation of Local Tissue Reaction After the Application of a 3D Printed Novel Holdfast Device for Left Atrial Appendage Exclusion,’ researchers from Poland begin to experiment even further with 3D printed devices for assistance in cardiac procedures. Here, they experiment with the use of a new holdfast device meant ultimately to help eliminate chances of ischemic stroke. Employing the benefits of SLS 3D printing—with the capabilities to use versatile materials with even greater affordability—the researchers evaluated the results of their new device, implanting the devices in a group of 30 swine.
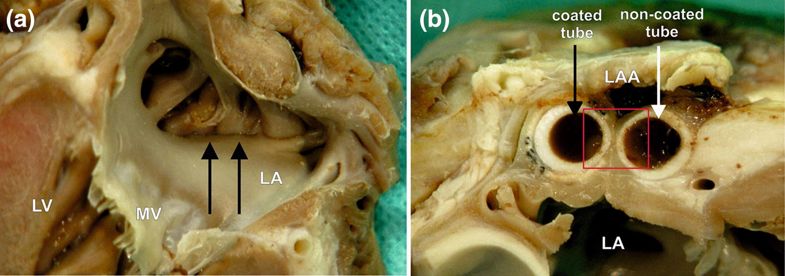
A macroscopic view of the left atrial appendage (LAA) closed with the holdfast device in group II (one tube coated with vascular implant). (a) The inner surface of the left atrium (LA) and (b) the intersection through the area of a holdfast device is shown. (a) Black arrows indicate the line of atrial wall adhesion placed between tubes of holdfast device. (b) The white arrow indicates the tube of holdfast device without and black arrow with a vascular implant. Red square indicates area showed in Fig. 8. MV mitral valve, LV left ventricle.
A finger-like extension of the left atrium, the left atrial appendage is a non-functional part of the left atrium. It happens to be a ‘prime cardiac site’ for thrombus formation, affecting patients with atrial fibrillation more commonly. And while treatment with antithrombotic treatment is widespread, here, the researchers investigate a more comprehensive approach in ‘obliterating the LAA.’ The use of holdfast devices, while effective, has been fraught with disadvantages, from issues with blood leakage to post-insertion adjustments, and lack of flexibility for modifications.
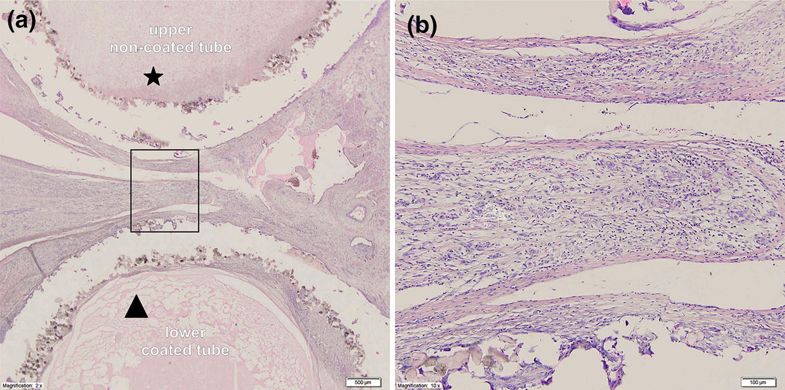
Microscopic view of the intersection through a holdfast device coated with a vascular implant used in group II. (a) Cross section through the device and the area between tubes of the holdfast device in group II. The the upper tube (*) was not coated with vascular implant and polyamide powder could be seen with an area of cicatrization. The lower tube (triangle) is coated with vascular implant and significant chronic inflammatory infiltrate is visible. (b) Higher magnification of the studied area with visible large number of giant cells.
“Considering the advantages and disadvantages of the available LAA closure devices, we designed a novel holdfast device for LAA obliteration devoid of these shortcomings,” stated the researchers.
The study allowed the researchers to further test biocompatibility, along with observing post-implementation issues. The device was created in partnership with the Technical University of Gdańsk, Poland, made up of two tubes connected with an elastic bow.
“The concept of operation for the device was based on the idea that the tubes would press the LAA base, which would lead to its closure and occlude the connection between the LAA and left atrium,” stated the researchers. “Moreover, the intention was that the device could be freely positioned around the LAA and could be easily adjusted after implantation.”
Their goal was to create a low stiffness spring with a ‘highly degressive characteristic of the device.’ They were able to do this through their choice of material and through shifting the bow crossection axis, with the devices implanted into the two swine groups. Safety and biocompatibility were noted, along with biofunctionality.
There were no perioperative deaths, and LAA occlusions occurred in all the swine.
“The foreign body reaction of the LAA holdfast device made of polyamide powder was found to be at a statistically insignificant level and was lower when compared to the polyester graft. Our device fulfills the criteria of biocompatibility to the highest degree, which makes it a suitable material for manufacturing LAA holdfast devices,” concluded the researchers. “This study proves that our technology may be applied for quick, innovative and individualized production of medical implants.“
3D printing has made huge strides in the medical realm, and definitely within the area of cardiac medicine, from cardiac patches to catheter devices, and even patient-specific heart valves models. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source / Images: ‘Evaluation of Local Tissue Reaction After the Application of a 3D Printed Novel Holdfast Device for Left Atrial Appendage Exclusion’]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Nikon SLM Solutions Sells SLM 500 to Primary Weapon Systems to Expand Suppressor Production
Primary Weapons Systems (PWS) is a Boise, Idaho-based manufacturer of suppressors, firearms, and related components. A subsidiary of Vigilant Gear and a sister company to aftermarket Glock slide manufacturer Lone...
3DPOD 261: Tooling and Cooling for AM with Jason Murphy, NXC MFG
Jason Murphy´s NXC MFG (Next Chapter Manufacturing) is not a generalist service; instead, the company specializes in making tooling. Using LPBF and binder jet, the company produces some of the...
HP and Firestorm Labs Form Partnership to Use Multi Jet Fusion 3D Printers in Deployable Factories
HP Inc., maker of a range of additive manufacturing (AM) solutions including the Multi Jet Fusion (MJF) ecosystem, has announced a partnership with Firestorm Labs, a developer of containerized, deployable...
3D Printing News Briefs, July 2, 2025: Copper Alloys, Defense Manufacturing, & More
We’re starting off with metals in today’s 3D Printing News Briefs, as Farsoon has unveiled a large-scale AM solution for copper alloys, and Meltio used its wire-laser metal solution to...