 Pioneer bioprinting company Organovo presented new findings in their stem-cell-based approach to developing kidney tissue using its 3D bioprinting platform, the NovoGen Bioprinter. During the annual meeting for The International Society for Stem Cell Research, held during the last week of June in Los Angeles, California, the company presented new data that demonstrates the automated production of complex kidney organoids, with potential future applications including in vitro disease modeling and the treatment of patients with renal disease.
Pioneer bioprinting company Organovo presented new findings in their stem-cell-based approach to developing kidney tissue using its 3D bioprinting platform, the NovoGen Bioprinter. During the annual meeting for The International Society for Stem Cell Research, held during the last week of June in Los Angeles, California, the company presented new data that demonstrates the automated production of complex kidney organoids, with potential future applications including in vitro disease modeling and the treatment of patients with renal disease.
“Partnerships with world-class institutions can accelerate groundbreaking work in finding cures for critical unmet disease needs and the development of implantable therapeutic tissues,” said Taylor J. Crouch, CEO of Organovo. “Our recently announced collaboration with Murdoch Children’s Research Institute (MCRI), in Melbourne, Australia, and Melissa Little (head of the Kidney Research laboratory at MCRI) has made our work automating the fabrication of kidney organoids possible. By combining MCRI’s proprietary approach for modeling human kidney tissue from stem cells and Organovo’s 3D bioprinting platform, we’re able to produce detailed kidney tissues, which is a key step toward advancing this promising technology for both drug testing and therapeutic applications. We’re hopeful that this will be an important step along the way in treating kidney disease.”
According to Organovo publications, recent advances in the directed differentiation of human pluripotent stem cells (also known as human embryonic stem cells) to kidney organoids advances the prospect of drug screening, disease modeling, and even restoration of renal function using patient-derived stem cells.
Single-cell transcriptional profiling shows the equivalence between standard and bioprinted kidney organoids
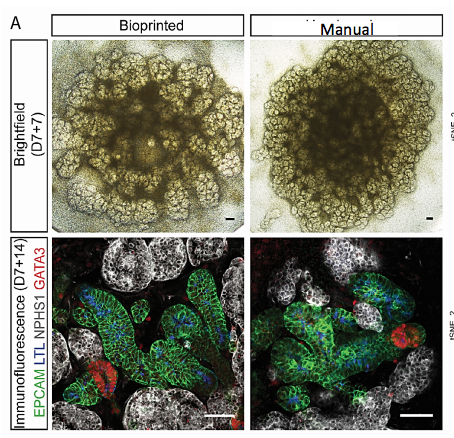
They claim to have demonstrated the successful adaptation of the directed differentiation protocol on the NovoGen Bioprinter MMX technology to achieve automated, rapid fabrication of self-organizing kidney organoids. Bioprinted organoids were found to be equivalent to those previously reported via manual generation, at both the level of morphology and component cell types and expression profiles. High-throughput toxicity screening was achieved by treating organoids bioprinted in 96-well plates with a classic nephrotoxic compound. Collectively, these results suggest that bioprinted kidney organoids are functionally equivalent to those prepared manually and thus are likely to be useful for a multitude of applications. In fact, using a bioprinter allows for the generation of large numbers of uniform and highly reproducible organoids in reduced time (approximately 20 times faster) compared to manual processes.
So 3D bioprinting enables automated and scaled fabrication of human-induced pluripotent stem cell (iPSC)-derived kidney organoids equivalent to those generated manually at the level of cellular complexity, identity, and gene expression. In addition, the inclusion of the bioprinter increased speed and reproducibility facilitating larger production runs without comprising organoid quality. The work is significant to the utility in drug testing and modeling human development and disease in vitro and provides translational promise for the combined use of iPSC and tissue engineering technologies for functional restoration in patients with renal disease.

Characterization of kidney organoids bioprinted using control and reporter iPSC lines, showing evidence of increasing tubular complexity (scale bar represents 800 µm)
Companies around the world are heavily pushing the limits of bioprinting to get closer to tangible and robust results that will prove useful to patients worldwide in just a few years. Finding solutions for conditions like renal failure and disease are on the top of the list for many bioprinting companies and researchers. The Global Burden of Disease (GBD) estimated in a study that in 2015 alone, 1.2 million people died from kidney failure, an increase of 32% since 2005. In 2010, between 2.3 and 7.1 million people with end-stage kidney disease died without access to chronic dialysis. Additionally, each year, around 1.7 million people are thought to die from acute kidney injury. Overall, therefore, between five and ten million people may die annually from kidney disease.
The San Diego-based biotech firm has been targeting 3D bioprinted tissues with human functionality for over 10 years, pursuing IND-track programs to develop its NovoTissues to address a number of serious unmet medical needs, initially focusing on liver disease. In 2017, Organovo’s program for Alpha-1-antitrypsin deficiency received orphan drug designation from the FDA and they have also provided access to its ExVive in vitro tissue platform to facilitate high-value drug discovery and development collaborations. As part of their hopes to advance medical care, Organovo has focused much of their research on kidney tissue, releasing a plethora of information regarding the performance of its 3D printed kidney tissue in research and testing over the last five years. It’s all part of the advances that are paving the way for the future of regenerative medicine and personalized patient care, two of the biggest challenges humanity faces with an aging population, which continues to grow in numbers and evolving healthcare needs. This type of medical advance can help save lives and reduce the considerable costs of healthcare everywhere.
[Images: Organovo]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Portsmouth Naval Shipyard Installs 3D Printed Part on In-Service Submarine in Key Milestone for US Defense Sector
I’ve been tracking the US Navy’s additive manufacturing (AM) buildup as it relates to submarines for a while now, and even amidst the AM efforts that all the branches have embarked...
6K Additive Lands Long-Term Nickel-Alloy Powders Supply Agreement with Siemens Energy
We’ve still got a month left in Q1, but I’ve already come back to the theme of the role of additive manufacturing (AM) in the ‘sustainability-as-security’ thesis — a topic...
Cheap Printers Make Metal Powders Costly
Last year, on stage at AM Strategies, the argument was simple: additive manufacturing had to stop chasing elegance and start chasing economics. The 3D printing community had spent decades refining...
Adoption of Advanced Powder Metal Manufacturing in the Global Small Arms Space – SHOT Show 2026 Additive Manufacturing Analysis
Back in 2013, I remember being completely blown away when a company called Solid Concepts reportedly additively manufactured nearly a complete firearm as a proof of concept. Although desktop polymer...

































