Self Healing Dynamic Polyurea Materials Show Significant Potential in 3D Printing
International researchers have been working together to improve on self-healing polymers, outlining their success in creating a covalent urea bond in the recently published ‘Dynamic covalent urea bonds and their potential for development of self-healing polymer materials.’ In a world of smart devices, smart materials make the ultimate sense for the longevity of parts, electronics, and more. Self-healing polymers are equipped with the ability to repair defects before the integrity of a product is harmed.
It is easy to see why industrial users—and ultimately, users on any level—would be interested in self-healing materials due to the potential for savings on the bottom line and extended performance. This type of value could be beneficial to nearly any application.
The authors explain that their focus turned to the urea bond due to its ‘pinnacle of stability’ and dynamic nature brought on through the mediation of zinc salts. Self-healing features could also help overcome some of the greatest challenges with polymers that usually cannot be repaired after they become damaged. This is an obvious environmental concern, along with an economical one. Repairable polymers currently are sustained through the following:
- Reversible or irreversible covalent bonds
- Supramolecular interactions
- Chemo-mechanical features
- Shape memory
“In addition, recent studies have also shown that van der Waals forces as well as microphase separation in thermoplastic polymers can also facilitate self-healing without elaborate chemical modifications,” state the researchers. “Among these methods, the use of dynamic covalent bonds to prepare self-healing polymer materials is widely studied, since no external event needs to take place, and the self-healing process can be repeated several times.”
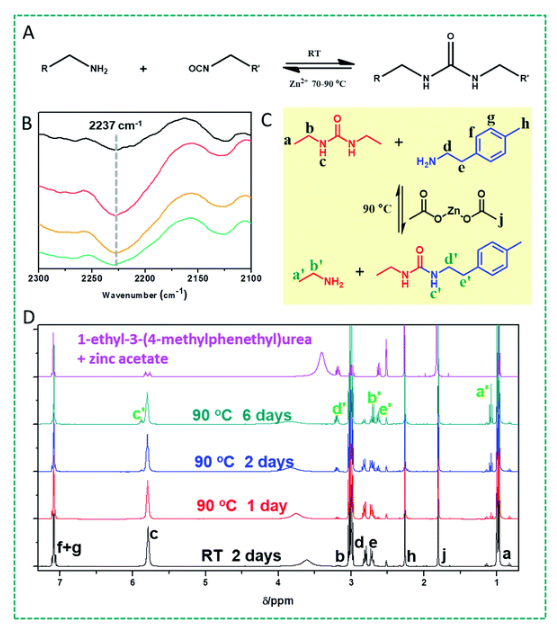
(A) Fundamental question of this study: can the urea bond be made reversible by Lewis acid catalysis in the presence of zinc ions at different temperatures, and thus open up the way for intrinsically self-healing polyureas? (B) FTIR spectra of a linear PDMS–Zn(OAc)2: a freshly prepared sample (black curve, top); the fresh sample was heated at 90 °C for 16 h (red curve), and then cooled down to room temperature for 24 h (yellow curve), and finally kept at room temperature for 72 h (dark cyan curve, bottom). (C) Reaction between 1,3-diethylurea and 2-(p-tolyl)ethylamine in the presence of Zn(OAc)2. (D) 1H NMR spectra of a 1,3-diethylurea, 2-(p-tolyl)ethylamine and Zn(OAc)2 mixture (reaction shown in (C)) reacted in DMSO at room temperature for 2 days (black curve), and at 90 °C for different time periods (red, blue and dark cyan curves); the pink spectrum (top) is that of a mixture of independently prepared 1-ethyl-3-(4-methylphenethyl)urea and zinc acetate.
Self-healing polymers have been created previously in relation to dynamic covalent chemistry, mainly through ‘the condensation-polymerization of a chain extender, cross-linker and small molecules containing dynamic covalent bonds.’
In adding other dynamic bonds, however, the chemicals become complex enough to transform the properties in a negative fashion.
“In contrast, if one is able to impart dynamic characteristics to conventional chemical bonds that are used for chain extending and cross-linking, and achieve self-healing functions with those very moieties, then that would be of great importance since no additional external bonds need to be introduced into the polymer networks,” state the researchers.
Here, they endeavored to transform the urea moiety into a ‘dynamic covalent motif,’ attempting to develop the desired materials via transcarbamoylation processes. They studied reaction kinetics both with and without Zn(OAc)2 by:
- Nuclear magnetic resonance (NMR)
- Fourier-transform infrared spectroscopy (FTIR)
- Gas chromatography-mass spectrometry (GC-MS)
The team further confirmed ‘dynamic properties’ of the urea bond through a series of ‘quantum chemical density-functional theory calculations.’ Afterward, they forged ahead in creating materials, preparing two different polydimethylsiloxane–Zn(OAc)2 (PDMS–Zn(OAc)2) cross-linked polymer networks and examining them, noting that the presence of zinc ions accelerates dissociation of ureas by ‘two orders of magnitude.’
“Relying on this dynamic urea bond, we developed self-healing Zn(OAc)2-containing polymeric materials that, upon mechanical scratching, can be fully repaired via a simple thermal treatment,” concluded the researchers. “Due to the wide range of commercially available primary amines and isocyanate monomers, this method can be used to prepare many kinds of novel self-healing and reprocessable polyurea materials.”
“As a result, dynamic polyurea materials are expected to have significant potential in a range of applications, such as coatings, paints, and 3D printing. Studies on the influence of different catalysts on the dynamic character of urea bonds are currently ongoing in our labs.”
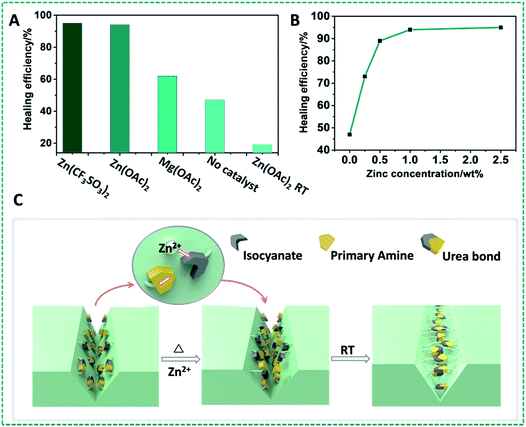
A) Healing efficiency of PDMS materials with different catalysts at 90 °C. (B) Healing efficiency of the PDMS–Zn(OAc)2-I network (P4) as a function of Zn(OAc)2 concentration. (C) Schematic illustration of the self-healing process of the PDMS–Zn(OAc)2 networks: in the presence of Zn ions, urea moieties can open up and subsequently restore the materials by re-forming in novel geometries.
If you have taken an interest in 3D printing, chances are you have taken an ever-expanding interest in materials too, as the choices continue to grow, and manufacturers and researchers continue to improve on polymers, from the use of grayscale to silicone resin to other plant-based derivatives. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
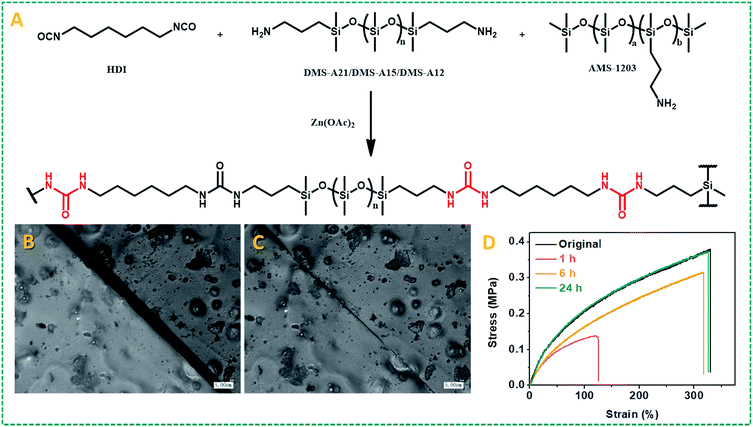
(A) Schematic representation of the synthetic route of the PDMS–Zn(OAc)2-I network. (B) Optical image of the as-prepared PDMS–Zn(OAc)2-I film (P1) with a scratch (scar bar: 5 μm). (C) Optical image of the damaged PDMS–Zn(OAc)2-I film after thermal treatment at 90 °C for 30 min (scar bar: 5 μm). (D) Stress–strain curves of the original PDMS–Zn(OAc)2-I (P1) films and the cut-off films after thermal treatment at 90 °C for different time periods.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Astrobotic Tests Rocket Engine Made with Elementum 3D Materials
Astrobotic has completed a series of hot-fire tests for its Chakram rotating detonation rocket engine, with additive manufacturing (AM) playing an important role in how the engine was built. The...
ARC & ORNL Form Partnership to Accelerate AI-Enabled Manufacturing for Defense
Last year, Autonomous Resource Corporation (ARC) became the surprising owner of Desktop Metal’s (DM’s) assets following the bankruptcy of the one-time additive manufacturing (AM) unicorn, an acquisition that cost ARC...
HP Stock Jumps on 3D Printing Buzz
HP (NYSE: HPQ) had its best day in over a year this week, with shares jumping more than 7% on Tuesday. Interestingly, the move was quickly tied to 3D printing,...
HP’s MJF 1200 Targets Entry-Level AM — And Could Shift the Competitive Landscape
HP has launched an entry-level MJF solution, the HP Multi Jet Fusion 1200 3D Printer Solution, which will be available in 2027. What is the 1200 exactly? What does it...










































