Interview with Shane Quinn of AddiHive On Serial Manufacturing Through 3D Printing
We got the chance to interview Shane Quinn of AddiHive, an Ireland based 3D printing company. We covered the firm before, mentioning how they wanted to bring serial production to medical device and aerospace in 3D printing. This is a particularly challenging area to work in. On the one hand, it is an area where firms could use a lot of aid, advice, and technology. On the other hand, is the young Limerick based firm up to the challenge? Working in highly regulated environments with some of the tightest requirements and tolerances would require enormous dedication, precision, and operational excellence. Bringing the, almost but so totally not completely, ready for production 3D printing technology to a playing field where everyone eats ISO soup and GMP is a jumping off point will be quite the challenge. The beauty of AddiHive though is that from a competitive standpoint they would be extremely difficult to dislodge once they do get their proposition right. AddiHive started with powder management but now wishes to do much more, we asked Quinn to tell us what.

What is AddiHive?
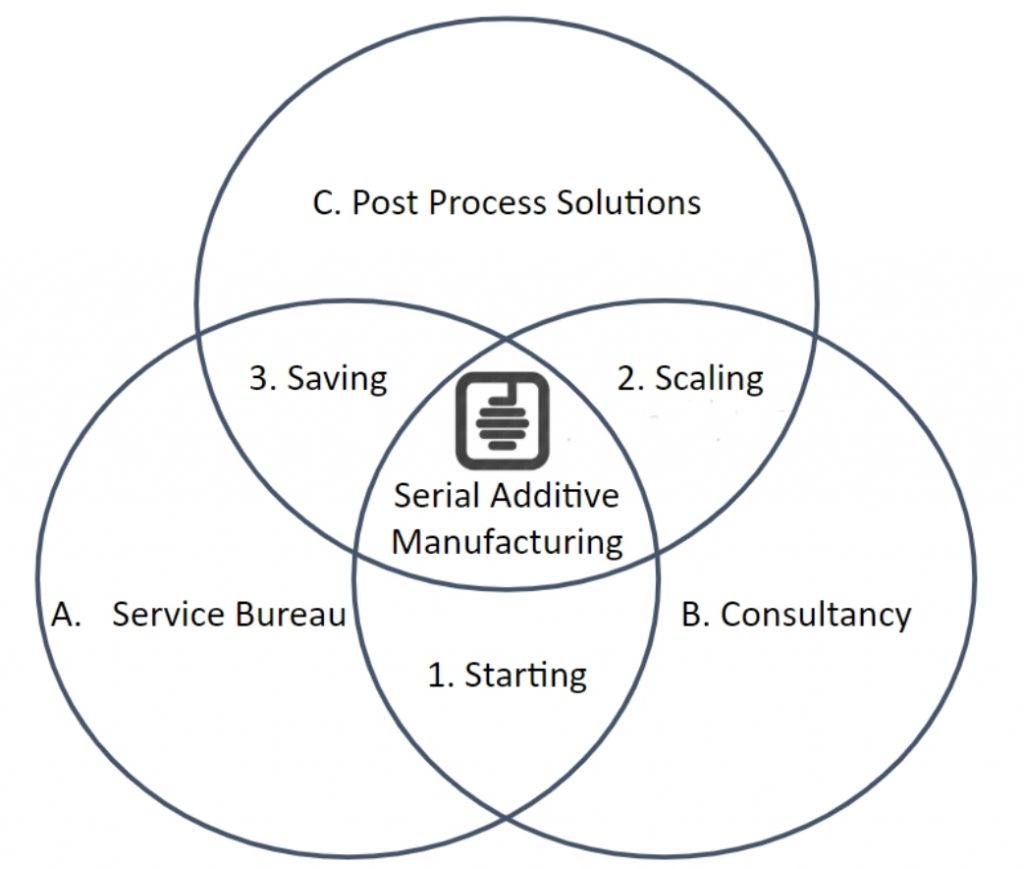
AddiHive is a Serial Additive Manufacturing Solutions Provider. Our customers are AM part manufacturers.
AddiHive serves the 3 key customer life-cycles:
1. Starting (customers starting out in AM with a view to scaling to serial AM)
2. Scaling & Stabilisation (customers that have an AM process and want to scale it & stabilise it effectively – move to serial AM)
3. Saving (This is a business, Cost Saving Pressures are inevitable once a serial AM process is scaled and stabilised)With 3 key services:
1. AM Service Bureau Prototyping, Subcontract Manufacture including Post Processing, Ancillary Services & Solutions (eg. Machine Setup Service and Equipment, Troubleshooting Equipment, Process Control Equipment, Powder Mix avoidance equipment…)
2. AM Consultancy & Contract AM Engineers, On Demand Consultancy
3. AM Post Process Solutions Powder Loop, Powder Clean & Powder Management System
Why is it so difficult to make serial parts?
1. End to End Process Variation
The AM build process has variation (part to part, build to build, machine to machine and machine type to machine type). Every other step of the AM process also has variation (Part removal, Support removal, Powder Removal, Heat Treatment…) & this variation quickly stacks up. In serial AM it becomes incredibly important to setup your machines to tight tolerances and accurately control variation.2. Lack of Key AM Variables Understanding
It’s hard to find people with serial AM experience. But its vital to have the right people on board, who understand the pitfalls, to scale efficiently.
What advice would you give to a company that wishes to manufacture using 3D printing?
Build a strong foundation at the start by getting the right advice (talk to people that have experience of scaling). You can build a strong foundation by:
1. Choosing Equipment & Software solutions and partners carefully
2. Minimising equipment variation from the start (1. part to part, 2. build to build, 3. machine to machine and 4. machine type to machine type) with a watertight setup and control of the process.
3. Having a firm grasp of key process variables & controlling these variables tightly.
4. Having a lean and smart QC and validation strategy to enable future cost-cutting.Understand that Serial Additive Manufacturing is different to Additive Manufacturing. With scale comes variation & controlling this is paramount.
Its relatively easy to build 1 part but building 1000 parts, considering variation between parts, builds, machines & machine types, that’s a different ball game.
What advice would you give to a medical device manufacturer?
1. Understand all of the AM specific FDA technical considerations inside out and align your AM QC and AM validation strategy to enable future changes (cost cutting pressures will come). Notice how much more digitised the manufacturing process is compared to conventional manufacturing.
2. Powder removal is probably one area you want to focus a lot of energy. Powder Removal tends to be a higher focus for medical device manufacturers (Lattice structure =Powder Removal).
3. Maintain a constant R&D mindset, bank R&D studies & information so they can be called upon when needed, don’t wait for issues to occur before digging deeper into the detail.
What kinds of aerospace parts do you want to help get made?
We specialise in the field of serial AM. So, any parts that requires large volumes and minimal variation (1. Part to part 2. Build to build 3. Machine to machine and 4. Machine type to machine type.)
Who do you partner with?
1. AddiHive is a spin out of Croom Precision Medical. We have worked extremely closely with them to develop their AM process from day one.
2. Our more famous partners prefer to remain anonymous. We don’t mean to be rude but we respect their desire to anonymity.
Who produces the parts?
We partner with Croom Precision Medical to product AM parts. We produce these parts in an FDA Registered facility with ISO 13485:2012, ISO9001, ISO 14001 accreditation & Japanese PMDA.
However AddiHive offers much more than just AM parts, we are an AM solutions provider, our aim is to enable AM part manufacturers like Croom Precision Medical to Start, Scale & Save.
What is Powder Loop?
Powder Loop is:
- a raw material recycling system that recycles reactive metallic powder in the same time it takes to clean an additive manufacturing machine.
- It eliminates the need for a stand alone sieve,
does not compromise floor space and- it eliminates the risk of powder mix up.
Ultimately it maximizes the productivity of your additive manufacturing machine, which reduces your cost/ part.
The quality of your raw material and material life-cycle can be managed centrally by connecting multiple Powder Loops to a central Powder Management System.

What can data help us do in 3D printing?
1. Data can reveal any unknowns with the process. With Statistical Process Control you can forsee and intervene before an issue occurs.
2. Ultimately data can provide a machine learning capability that will enable machines to correct themselves to maintain desired process parameters.
What is holding 3D printing back?
1. Raw material lifecycle visibility and management (powder quality control, risk of powder mix ups)
2. The need for operator intervention (manual tasks) Manual = variation and cost.
3. unpredictable outcomes (process simulation software is attempting to solve this)
With many variables on the end to end process you often require many product iterations to predict outcomes.
Why is robotics important?
Robotics & automation will reduce variation and ultimately cost/ part (in the same way self driving cars will reduce accidents).
What kinds of cost savings could you achieve?
1. Service Bureau
*Close control of Machine Setup and effective Troubleshooting will minimise scrap and increase productivity.
We can confidently guarantee payback on all setup equipment within the 1st year of production (reduced scrap)2. Consultancy
The cost saving on consultancy is very project dependent, all we can say is:
*What is foreseeing and avoiding a significant problems worth? Our End to End process knowledge allows us to provide solutions to a variety of issues.3. Post Process Solutions (powder loop)
Obviously it depend on the AM machine used & parts built, but as an example, if you say your laser to laser time is currently 20 hours
*we’re confident we can reduce that by 2 hours (10%). So you can produce more parts with less machines.
*The value of material mix avoidance cannot be understated. The Powder Loop eliminates this risk.
What are you doing in QA?
Our Powder Management System will offer an opportunity to monitor raw material chemistry (O2, N, H) in realtime, providing confidence in raw material quality from cradle to grave.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Amnovis Expands to the US
Belgian firm Amnovis is a scalable partner for orthopedic innovations. From design to production and regulatory, you can rely on them to take your innovation to market. Coupled with deep...
Scaling 3D Printing Takes More Than You Think; HP’s Webinar Looks at Making It Work Long Term
3D printing is no longer new. In fact, most manufacturers already know what it is, where it fits, and what it can do. But knowing the technology and actually scaling...
Astrobotic Tests Rocket Engine Made with Elementum 3D Materials
Astrobotic has completed a series of hot-fire tests for its Chakram rotating detonation rocket engine, with additive manufacturing (AM) playing an important role in how the engine was built. The...
The Additive Chicken Coop, Part I: Million Dollar Petri Dishes
After decades of tinkering with our individual technologies, billions were poured into speculative claims and optimism. Now the attention is gone, and sometimes it can seem like we’re surrounded by...













































