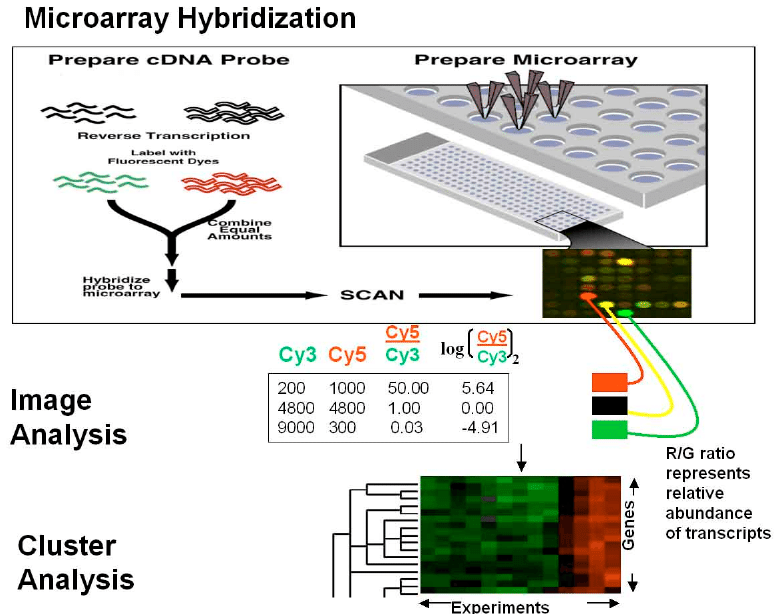
A DNA microarray can be referred to as a DNA chip or biochip. This technology refers to a collection of microscopic DNA spots attached to a solid surface. Scientists typically use microarrays to measure the expression levels of large numbers of genes simultaneously or to genotype multiple regions of a genome. So how is this technology relevant to bioprinting? We will go through this intriguing technology and how it affects bioprinting as a whole.
In terms of bioprinting, we must classify microarray bioprinting. Microarray bioprinting refers to printing cells encapsulated in hydrogels in a spatially addressable manner using automated liquid dispensing robots such as microarray spotters. An automated liquid dispensing robot refers to pipetting as well as pumping procedures done through machine automation. One can think of typical bioprinting setups with programmable syringe pumps. Microarray spotters refer to the robotic instruments used for making microarrays. This technology deposits (or “spots”) cDNA probes onto a microscope slide or other substrate. In genetics, complementary DNA (cDNA) is DNA synthesized from a single-stranded RNA (e.g., messenger RNA (mRNA) or microRNA) template in a reaction catalyzed by the enzyme reverse transcriptase. Cells are usually within the nanoliter volume (30 – 60nL) and can be dispensed onto functionalized glass slides or micropillar/microwell chip platforms. This is directly related to our previous article on DNA and how we talked about microfluidics. These cells can be grown in growth media to support miniaturized 3D cell cultures for toxicology assays. Toxicology assays are important in terms of bioprinting because one wants to make sure beforehand that the material being printed will not be detrimental or toxic to the body.
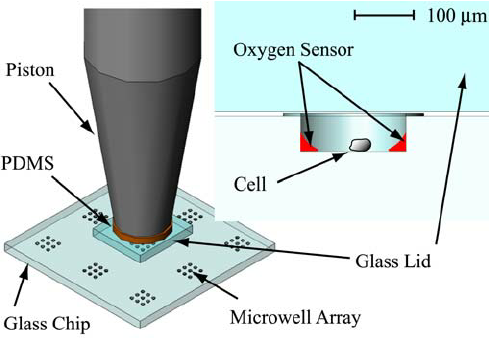 Microwell Chip and Extrusion setup
Microwell Chip and Extrusion setup
The microwell chip mentioned previously can accommodate up to 950 nL of compounds, recombinant viruses, growth factors, and fluorescent dyes for various cell-based assays. Since the micropillar chip is complementary to the microwell chip, cells on the micropillars can be exposed to hundreds of different test conditions in the microwells simultaneously by simply sandwiching the two chips together. The cells on the micropillar chip can be exposed to compounds for a period of time and stained with fluorescent dyes or fluorescently labeled antibodies to assess drug efficacy and toxicity. Compared to traditional 2D cell monolayer cultures, microarray bioprinting offers several attractive features, including physiologically relevant cells grown in 3D, miniaturization of cell-based assays saving valuable raw materials, such as primary human cells obtained from patients, and ultrahigh-throughput capability of testing cell culture conditions.
It is important to outline why physiologically relevant cell growth in 3D, miniaturization of cell-based assays, and ultrahigh-throughput capability of testing cell culture conditions are very important within microarray based bioprinting. In terms of microfluidics, the various cell structures in the human body must be vascularized to receive the nutrients and gas exchange help that they need to survive. Similarly, 3D cell cultures in vitro require certain levels of fluid circulation, which can be problematic for dense, 3D cultures where cells may not all have adequate exposure to nutrients. This is particularly important in hepatocyte cultures because the liver is a highly vascularized organ. Miniaturization of cell-based assays allows for rapid prototyping as well as low cost implementation within bioprinting processes. High-throughput screening (HTS) is a method for scientific experimentation especially used in drug discovery and relevant to the fields of biology and chemistry. Using robotics, data processing/control software, liquid handling devices, and sensitive detectors, high-throughput screening allows a researcher to quickly conduct millions of chemical, genetic, or pharmacological tests. Being able to do this in vitro allows us to feel more confident in the what is being used within the body in terms of bioprinting.
Microarray bioprinting is a promising high-throughput approach for miniaturized 3D cell cultures. The reduction in sample volume and the speed of printing hundreds of samples in a short period of time make this technology suitable for wide ranges of cell-based assays. The biggest issue is learning how to control these microenvironments enabled by microarray bioprinting.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
trinckle’s Tool Design Software Lands in Stratasys GrabCAD Print
I’m a fan of trinckle, the German startup that offers super easy-to-use tools for creating customized jigs, fixtures, and other 3D printed products. With trinckle, it’s not just designers who...
Caracol Taps CNC Robotics to Build and Support Its UK Systems
No matter how quickly the economy seems to be changing on the surface, there is no escaping the fact that geography is the foundation of economics, and, in case anyone...
EOS Buys Metalpine, but What’s Behind the Move?
EOS has bought Austrian powder manufacturer Metalpine. EOS doesn’t buy companies often. And with increased competition from China & SLM, profligate spending doesn’t seem like it would be a good...
ExOne Cuts Costs for U.S. Customers as Printhead Production Moves to Detroit
ExOne Global Holdings, created through the 2025 integration of ExOne and voxeljet, is making changes across its U.S. operations. These include starting printhead manufacturing in the Detroit area and lowering...