Oklahoma State University Dissertation: Bioprinting Neural Tissue with Chitosan-Gelatin Hydrogels
 Kevin Roehm of Oklahoma State University is investigating the use of new materials and structures for successful bioprinting. His findings are outlined in his recently published dissertation, ‘Bioprinting Cell Laden Structures with Chitosan-Gelatin for Neural Tissue Applications.’
Kevin Roehm of Oklahoma State University is investigating the use of new materials and structures for successful bioprinting. His findings are outlined in his recently published dissertation, ‘Bioprinting Cell Laden Structures with Chitosan-Gelatin for Neural Tissue Applications.’
Bioprinting has gone far past the stage of offering ‘potential’ these days, with numerous 3D printed surgical implants helping patients go on to lead a good quality of life, and in some cases, even saving their lives. In the US, approximately one billion individuals are suffering from neurological conditions, and Roehm points out that there is a definite need for better tools to study difficult diseases like Alzheimer’s—as well as conditions like migraines. Roehm also explains that many surgeries are performed every year to repair nerves but that in neural tissue, ‘very little repair capability’ is found.
“Combined there is a significant need for synthetic, 3D tissues utilizing human cells for both in vitro models and regenerative applications,” says Roehm.
Typical challenges in bioprinting include material issues like finding or creating stable bioinks that are also stable under suitable conditions. Keeping cells alive is the other pressing issue, as there may be difficulty in stabilizing gels, managing operational settings, and shear stress. The author realizes a need for hydrogel gels that are crosslinked with the appropriate agents, and able to gel into a solid state:
“I propose using chitosan-gelatin -β-glycerophosphate (2GP) hydrogels, which exhibit thermogelation at physiological temperature (gelation occurs when the temperature is increased to 37 °C). My work focuses on exploring the printability and obtainable resolution of a hydrogel that has a unique gelation mechanism and does not require post-processing,” states Roehm.
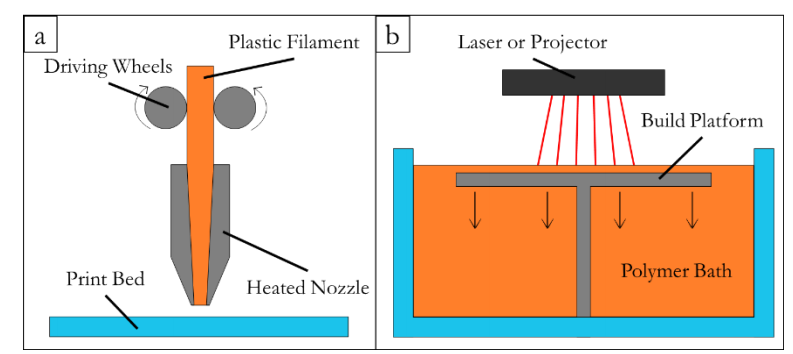
As Roehm points out, bioprinting is still faced with many challenges, and the actual technology used to print cells faces issues such as heat (FDM 3D printing kills the cells) and lack of overall viability (in use of SLS or SLA). Laser assisted bioprinting (LAB) is a choice also, but flow rate is usually too low, the technology is expensive, and the process is more inconvenient overall.
With inkjet printing, bioprinting is possible with hydrogels, but again there are issues with cell viability:
“This technology requires a crosslinking agent to solidify the printed material and inks must be non-viscous (less than 10 centipoise) prior to expulsion. Moreover, there is concern that the piezoelectric ejection systems used in inkjet printers might severely decrease cell viability. This problem is increased by the difficulty printing high cell density droplets. However, they are attractive because they are inexpensive and fast.”
Bioprinting is obviously an extremely complex process—especially when you consider that the end goal for so many scientists is to begin printing human organs in the lab. For success in bioprinting, however, inks must be non-toxic, and able to transform from a liquid to solid without threatening the sustainability of living cells. Hydrogels are intrinsically weak, but because they are so similar to soft tissues, the author points out that imitating their structure still results in greater success.
“Our hydrogel’s mechanical strength can be tuned to match tissue mechanical properties by varying the concentration of polymer and adding crosslinking agents such as hyaluronic acid and transglutaminase,” states Roehm. “Thus far, members of our laboratory have matched our hydrogel to both cartilage and cardiac tissue.”
The gelatin-chitosan-β-glycerophosphate hydrogel has proven itself to be ‘conducive for adhesion of various cell types,’ and does not require post processing. The researchers have discovered that both stability and mechanical properties can be manipulated as needed by cross-linking gelatin. CG structures showed ‘high viability’ even after five days.
One bioprinter used in this study was a motion platform kit donated by Maker’s Tool Works (Oklahoma City), with custom Python code written for modeling; however, Roehm discovered that his low-cost, customized printer was perfect for the job:
“My results show that my printer is suited to initial evaluation of novel inks where a more expensive system represents too much risk. Our CG hydrogel has not previously been investigated as a bioprinter ink and did not justify significant investment in technology without evaluation of its suitability as an ink. Our system allowed that evaluation to be conducted without risky investment in more expensive technology.”
Roehm points out that while most bioprinting research today focuses on sustainability and viability in cells, not enough focus has been placed on cell response as a different area of examination.
“However, there is very limited understanding of how bioprinting alters cell response; some assume that high viability infers normal function and unaltered stimulus response. Moreover, most inks require post-processing steps to stabilize the ink that may damage cells or alter their function,” concludes Roehm. “This work demonstrates a hydrogel that does not require post-processing and indicates that high viability does not imply unaltered stimulus response.”

Bioprinting parts description: a) Complete cell printing setup for printing of cell laden ink. b) CAD assembly of bioprinter. c) Custom print head and d) close up of syringe.
If you are interested in finding out about other research projects involving bioprinting and the use of hydrogels, check out other topics that touch on studies in 4D printed, shape-shifting structures, structures like injectable composites, and other innovations like smart hydrogels that are mobile. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.

Slicing a model. The initial 3D model (a), visual depiction of material deposited for
the first layer of the model as described by autogenerated G-Code (b), and a visual representation
of the 247,000 lines of G-Code generated to build the part (c).
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
RAPID 2026: 6K Additive’s Domestic Metal Powders & Consolidation Plan
6K Additive (ASX: 6KA), a U.S. supplier and manufacturer of metal powders for additive manufacturing (AM), has been very busy lately. I caught up with CEO Frank Roberts and Chief...
3D Printing News Briefs, April 22, 2026: DINOs, Post-Processing, AM for Aerostructures, & More
We’ll start with event news in today’s 3D Printing News Briefs, as AMUG presented its DINO Award to six members at this year’s conference, and Axtra3D celebrated its five-year anniversary...
Medical, Electronics, & Semiconductors: Detailed 3D Prints at RAPID 2026 with Boston Micro Fabrication & Lithoz
They say that good things come in small packages, and that’s certainly the case when it comes to Boston Micro Fabrication (BMF). A leader in micro-precision additive manufacturing, the company...
Bambu Lab’s 3D Printed Drone Add-On Helps Measure Whales from the Sky
Bambu Lab has helped Tandem Ventures develop the WHASER, a 3D printed attachment for a drone that can measure whales. This seems like a nifty tool for researchers. It is...