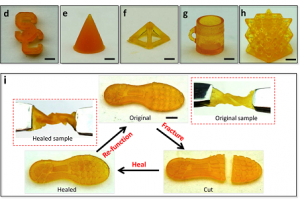
3D printed shoe soles were cut in half and self-healed in about 2 hours. [Image: An Xin and Kunhao Yu]
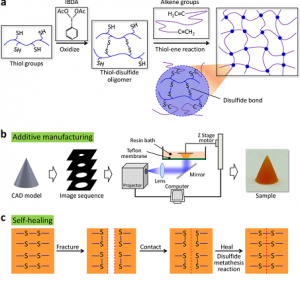
To make this material, the researchers use a 3D printing method that employs photopolymerization, which solidifies a liquid resin into a desired geometry or shape using light. The researchers further explain their work in a paper, titled “Additive manufacturing of seal-healing elastomers,” which was recently published in the journal Nature.
The abstract reads, “Nature excels in both self-healing and 3D shaping; for example, self-healable human organs feature functional geometries and microstructures. However, tailoring man-made self-healing materials into complex structures faces substantial challenges. Here, we report a paradigm of photopolymerization-based additive manufacturing of self-healable elastomer structures with free-form architectures. The paradigm relies on a molecularly designed photoelastomer ink with both thiol and disulfide groups, where the former facilitates a thiol-ene photopolymerization during the additive manufacturing process and the latter enables a disulfide metathesis reaction during the self-healing process. We find that the competition between the thiol and disulfide groups governs the photocuring rate and self-healing efficiency of the photoelastomer. The self-healing behavior of the photoelastomer is understood with a theoretical model that agrees well with the experimental results. With projection microstereolithography systems, we demonstrate rapid additive manufacturing of single- and multimaterial self-healable structures for 3D soft actuators, multiphase composites, and architected electronics. Compatible with various photopolymerization-based additive manufacturing systems, the photoelastomer is expected to open promising avenues for fabricating structures where free-form architectures and efficient self-healing are both desirable.”
 In making their material self-healing, the team had to do some in-depth research into its chemistry. You can achieve photopolymerization through a reaction with the thiols chemical group, which can transform into disulfides with the addition of an oxidizer. Objects made with this second group of chemicals are able to reform themselves if they break, so the researchers just needed to figure out the correct ratio.
In making their material self-healing, the team had to do some in-depth research into its chemistry. You can achieve photopolymerization through a reaction with the thiols chemical group, which can transform into disulfides with the addition of an oxidizer. Objects made with this second group of chemicals are able to reform themselves if they break, so the researchers just needed to figure out the correct ratio.
USC Viterbi Assistant Professor Qiming Wang explained, “When we gradually increase the oxidant, the self-healing behavior becomes stronger, but the photopolymerization behavior becomes weaker. There is competition between these two behaviors. And eventually we found the ratio that can enable both high self-healing and relatively rapid photopolymerization.”
The team can 3D print a 17.5 mm square out of the material in just five seconds flat, and whole objects can be fabricated in about 20 minutes. For the purposes of their study, which was funded by both the National Science Foundation and the Air Force Office of Scientific Research Young Investigator Program, the team tested out this capability on several products, including an electronic sensor, a soft robot, and a shoe pad.
Once these 3D printed items had been cut in half, they completely healed themselves two hours later at 60°C, with the exception of the electronics, which took four hours to heal because of the electricity-transmitting carbon. By increasing the temperature, the repairs will take place even faster, and the objects will still retain not only their function but also their strength.
“We actually show that under different temperatures – from 40 degrees Celsius to 60 degrees Celsius – the material can heal to almost 100 percent. By changing the temperature, we can manipulate the healing speed, even under room temperature the material can still self-heal,” said Kunhao Yu, USC Viterbi student and the first author of the study.
This self-healing material could help lower the manufacturing time, and increase durability and longevity, for many products in all sorts of industries.
The researchers are now focusing on making self-healing materials along a range of stiffnesses, from soft rubber to rigid plastics, which could one day be used to repair composite materials, body armor, and vehicle parts.
Co-authors of the paper are Yu, USC Viterbi students An Xin and Haixu Du, University of Connecticut Assistant Professor Ying Li, and Wang.
Discuss this research and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
[Source/Images: USC Viterbi]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing News Briefs, April 13, 2024: Robotics, Orthotics, & Hypersonics
In 3D Printing News Briefs today, we’re focusing first on robotics, as Carnegie Mellon University’s new Robotics Innovation Center will house several community outreach programs, and Ugogo3D is now working...
Rail Giant Alstom Saves $15M with 3D Printing Automation Software 3D Spark
3D Spark has entered into a three-year deal with the rail giant Alstom. Alstom, a transport behemoth with annual revenues of $16 billion, specializes in the manufacture of trains, trams,...
Meltio Expands Global Reach with New Partnerships in the Americas and Europe
Spanish 3D printing manufacturer Meltio has expanded its sales network across the globe. With the addition of three new partners in the United States, Brazil, Argentina, and Italy, Meltio aims...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...