
The H1N1 virus. [Image via Wikipedia]
This flu was particularly shocking not only because of its scale but because of the fact that rather than resulting in the deaths of the young, old, and sick as is normally the case during flu outbreaks, it killed primarily healthy young adults. There are a number of theories as to why this flu was so particularly deadly and samples of it that were preserved in the bodies of victims who were found frozen were subjected to study at the time in order to attempt to unravel its secrets. While some investigation has proposed that the virus itself was particularly aggressive, other research has suggested that rather than a difference in the virus itself, it was a change in global circumstances as a result of WWI, such as malnutrition and crowding in prison camps, that allowed the normal flu virus to wreak such deadly havoc. A recent theory suggests the possibility that aspirin poisoning contributed to the high number of deaths, as there was a distinct spike after the surgeon general recommended large doses of aspirin as treatment and in the wake of the expiration of Bayer’s patent on the drug in which a multitude of other companies rushed in to produce, and profit from, the production of aspirin.
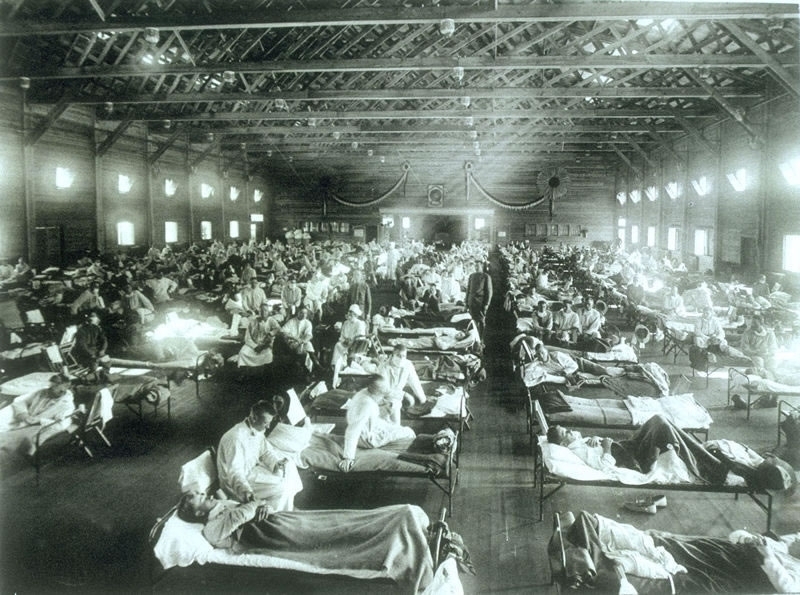
Soldiers from Fort Riley, Kansas, ill with Spanish influenza at a hospital ward at Camp Funston. [Image: US Military via Wikipedia]

On the left is a 1918 H1 influenza virus-like particle (VLP) as seen by cryo-electron microscopy. On the right is the same VLP rendered in 3D with structural components computationally segmented and colored; hemagglutinin and membrane are light blue and internal components (molecular cargo) are red. [Image: NIAID]
While it has been possible to get close up imagery of the VLPs before, never has it been presented with the kind of clarity made possible using 3D modeling techniques. This has enabled the researchers to understand the unique arrangement of hemagglutinin (HA) proteins and it is believed that the number and location of HA molecules may influence the efficacy of VLP vaccines. The hope is that with this increased understanding they will not only be able to create a potential platform for vaccines against other diseases, but also more efficacious vaccines against the flu itself, even when it’s just the regular seasonal bug.
The results of their preliminary research have been published in Scientific Reports, and will no doubt encourage others to understand the value that 3D modeling holds for medical research on this micro level. “Structural analysis of influenza vaccine virus-like particles reveals a multicomponent organization” was authored by Dustin M. McCraw, John R. Gallagher, Udana Torian, Mallory L. Myers, Michael T. Conlon, Neetu M. Gulati, and Audray K. Harris.
What do you think of this news? Let us know your thoughts; join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
[Source: NIAID]
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Unpeeled: New Arkema Material for HP, Saddle and Macro MEMS
A new Arkema material for MJF is said to reduce costs per part by up to 25% and have an 85% reusability ratio. HP 3D HR PA 12 S has been...
3D Printing News Briefs, January 20, 2024: FDM, LPBF, Underwater 3D Printer, Racing, & More
We’re starting off with a process certification in today’s 3D Printing News Briefs, and then moving on to research about solute trapping, laser powder bed fusion, and then moving on...
3D Printing Webinar and Event Roundup: December 3, 2023
We’ve got plenty of events and webinars coming up for you this week! Quickparts is having a Manufacturing Roadshow, America Makes is holding a Member Town Hall, Stratafest makes two...
Formnext 2023 Day Three: Slam Dunk
I’m high—high on trade show. I’ve met numerous new faces and reconnected with old friends, creating an absolutely wonderful atmosphere. The excitement is palpable over several emerging developments. The high...































