Wyss Institute: Bioprinted Kidney Subunits Offer Future Potential for Testing & Transplants
 Although 3D printing has truly earned its title as a revolutionary technology that will indeed change the way we do so many things worldwide—in nearly every industry—many have pointed out that change hasn’t come quite as quickly as they expected, whether that’s in the construction industry or manufacturing overall in the US and other countries. But such transformation certainly doesn’t happen in a day, or really even in a year. And while we hear of incredible strides within the medical field, the same holds true there as many wait for—and work to develop—the first 3D printed organ for transplant. That is the ultimate goal today in bioprinting, with predictions that while we might be able to create a 3D printed organ in the next year, actually transplanting one will probably take quite a bit more time.
Although 3D printing has truly earned its title as a revolutionary technology that will indeed change the way we do so many things worldwide—in nearly every industry—many have pointed out that change hasn’t come quite as quickly as they expected, whether that’s in the construction industry or manufacturing overall in the US and other countries. But such transformation certainly doesn’t happen in a day, or really even in a year. And while we hear of incredible strides within the medical field, the same holds true there as many wait for—and work to develop—the first 3D printed organ for transplant. That is the ultimate goal today in bioprinting, with predictions that while we might be able to create a 3D printed organ in the next year, actually transplanting one will probably take quite a bit more time.
As one would imagine, there are many steps involved in getting to the point where an organ, or even part of one, is bioprinted. That any part of the human body can be recreated in a lab is staggering in itself, and researchers continue to forge ahead with one successful step after another, building the way to their end goal of a transplantable organ. Now, researchers at the Wyss Institute at Harvard University have bioprinted their way to a functioning kidney subunit, as outlined in ’Bioprinting of 3D Convoluted Renal Proximal Tubules on Perfusable Chips’, by Kimberly A. Homan, David B. Kolesky, Mark A. Skylar-Scott, Jessica Herrmann, Humphrey Obuobi, Annie Moisan, and Jennifer A. Lewis.
In their paper, the research team—led by Core Faculty member Jennifer Lewis, Sc.D, who is also the Hansjörg Wyss Professor of Biologically Inspired Engineering at the Harvard John A. Paulson School of Engineering and Applied Sciences—explains just how they went about creating 3D printed proximal tubules that actually hold living cells and are able to mimic the duties of nephrons, which overall regulate substances like water and salt as they filter the blood. In successfully bioprinting these tubules, the team used what is called a fugitive ink. As you will see in the video below, it is liquefied and removed later, and then printed on top of an extracellular matrix.
This is not the researcher team’s first go round in bioprinting vascular tissues; in fact, they have not only done so previously, but they have created constructs that have been viable for over a month in vitro. This latest project from the Wyss Institute has been in collaboration with Roche Pharmaceuticals scientist Annie Moisan as the researchers create a 3D renal architecture that opens the door for a range of applications.
“The current work further expands our bioprinting platform to create functional human tissue architectures with both technological and clinical relevance,” said Lewis.
As they explain in their paper, the team combined bioprinting, 3D cell culture, and also organ-on-a-chip concepts as they worked to bioprint the complex architectures.
“These 3D convoluted PTs [proximal tubules] consist of an open lumen architecture circumscribed by proximal tubule epithelial cells (PTECs), embedded in an extracellular matrix, and housed within a perfusable tissue chip, where they are subjected to physiological shear stresses,” state the researchers in their paper.
The PTs are created as they print a silicone gasket on a glass slide to mark what is the outer border of the tissue chip. Next, engineered extracellular matrix (ECM) is placed within the gasket, and the fugitive ink is printed on top of that.
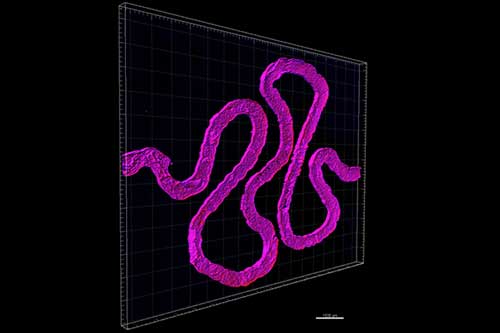
Fugitive ink is used to create bioprinted nephron tubules. [Image: Lewis Lab/Wyss Institute at Harvard University]
“After printing, the fugitive ink is connected to hollow metal pins interfaced through the gasket walls and additional ECM is cast over the printed structure. The 3D tissue model is then housed within a perfusable chip, where it is cooled to 4 °C to liquefy and subsequently remove the fugitive ink yielding an open convoluted tubular channel embedded within the ECM. Finally, cell media is perfused through the 3D convoluted tubular architecture on chip via an external peristaltic pump,” state the researchers.
“Notably, our method can create 3D proximal tubule models in myriad configurations with precisely controlled size, curvature, and location. For instance, if multiple tubules are required to increase statistical relevance of an assay or provide basal-side access channels, they can be printed alongside one another and either perfused independently or collectively through a single inlet.”
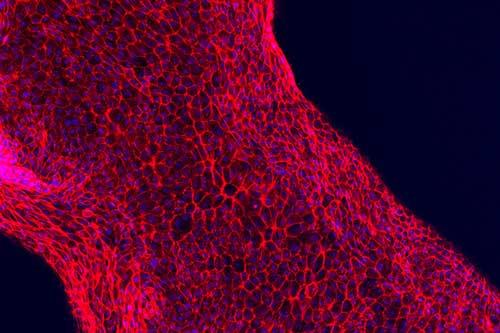
The scientists have been able to 3D print functional renal architectures. [Image: Lewis Lab/Wyss Institute at Harvard University]
“The use of functional tissue-like models during pre-clinical studies will provide unprecedented insights into human-relevant drug response prior to clinical development,” said Moisan, a Laboratory Head in Mechanistic Safety at Roche Pharmaceutical Sciences.
Their research doesn’t have to be limited just to kidney nephrons either. Due to the versatility of what they’ve created in the lab, the scientists will be able to translate their studies to that of other tissues and organs too.
“We have initially targeted this renal architecture, because the kidney represents such a pressing clinical need across the world,” said Lewis. “While thus far we have merely demonstrated a functioning subunit within the kidney, we are actively scaling up the method and its complexity to enable future in vivo applications.”
The team points out that with this new method, advances can be made as the living structures they create are conducive to drug testing by pharmaceutical companies, as well as advancing studies of disease models and propelling regenerative medicine further. Advanced tissue models on demand offer incredible potential for the medical field as a whole.
[Source: Wyss Institute]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Amnovis Expands to the US
Belgian firm Amnovis is a scalable partner for orthopedic innovations. From design to production and regulatory, you can rely on them to take your innovation to market. Coupled with deep...
Scaling 3D Printing Takes More Than You Think; HP’s Webinar Looks at Making It Work Long Term
3D printing is no longer new. In fact, most manufacturers already know what it is, where it fits, and what it can do. But knowing the technology and actually scaling...
Astrobotic Tests Rocket Engine Made with Elementum 3D Materials
Astrobotic has completed a series of hot-fire tests for its Chakram rotating detonation rocket engine, with additive manufacturing (AM) playing an important role in how the engine was built. The...
The Additive Chicken Coop, Part I: Million Dollar Petri Dishes
After decades of tinkering with our individual technologies, billions were poured into speculative claims and optimism. Now the attention is gone, and sometimes it can seem like we’re surrounded by...












































