If you or someone you know has sleep apnea, then you are probably also very aware that finding a long-term solution can be difficult—but one is much needed. This is a scary condition, and can be life-threatening too. Often accompanied by snoring that is loud enough to wake everyone in the house as well as some of the neighbors, sleep apnea is something for which most everyone wishes there was a magic solution. From sleep clinics to the traditional CPAP mask, treatments often seem available but difficult to keep up with—and are often left behind after a short time due to issues with comfort.
If you or someone you know has sleep apnea, then you are probably also very aware that finding a long-term solution can be difficult—but one is much needed. This is a scary condition, and can be life-threatening too. Often accompanied by snoring that is loud enough to wake everyone in the house as well as some of the neighbors, sleep apnea is something for which most everyone wishes there was a magic solution. From sleep clinics to the traditional CPAP mask, treatments often seem available but difficult to keep up with—and are often left behind after a short time due to issues with comfort.
Much of the problem, of course, is that with sleep apnea, patients are tired. They are tired all the time. When you snore, hold your breath, jolt awake, go back to sleep, and then repeat the whole cycle again over and over all night long, actual rest is elusive no matter how long you are in bed. And wearing an uncomfortable mask is often just not something those trying to get some rest can live with long term.
With hopes that the magic of 3D printing can come to the rescue here, Brisbane, Australia’s Oventus Medical has been working on a streamlined, compact solution in the form of their customizable titanium mouth device called the O2Vent. We’ve reported previously on Oventus and their collaboration with Australia’s Commonwealth Scientific and Industrial Research Organisation in bringing this innovative, customized breathing device to market in Australia this year.
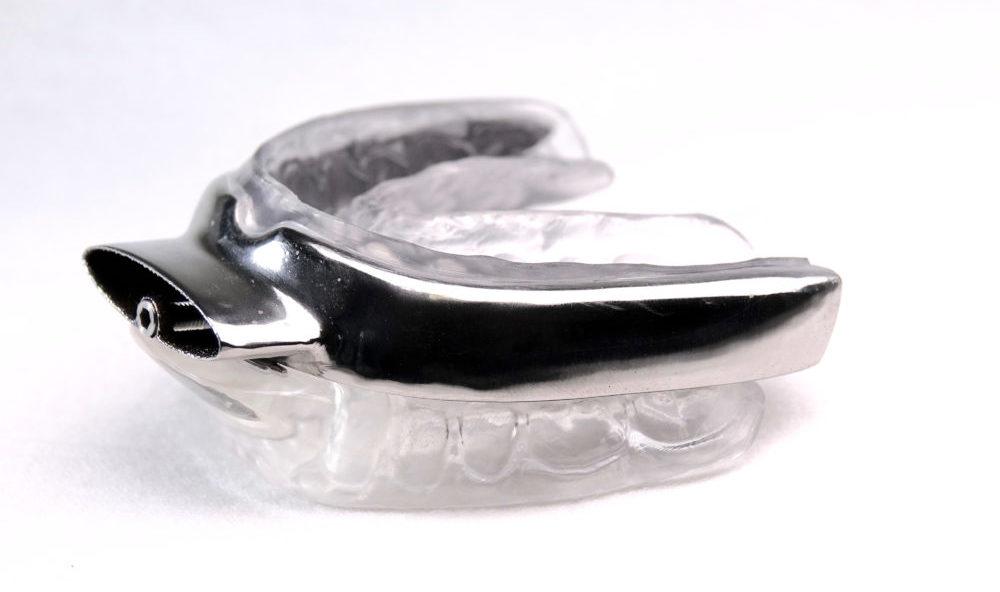
 The device is 3D printed in titanium. It’s compact and completely fitted to the individual’s mouth, directing airflow to the back of the throat and preventing obstructions. The Oventus team has created what they call a ‘duckbill,’ a portion of the device that allows for a separate airway pushing airflow to the back of the mouth. The O2Vent begins with a digital design of the patient’s mouth and bite and then is fabricated into a medical grade mouthguard.
The device is 3D printed in titanium. It’s compact and completely fitted to the individual’s mouth, directing airflow to the back of the throat and preventing obstructions. The Oventus team has created what they call a ‘duckbill,’ a portion of the device that allows for a separate airway pushing airflow to the back of the mouth. The O2Vent begins with a digital design of the patient’s mouth and bite and then is fabricated into a medical grade mouthguard.
Now, those suffering from sleep apnea are even closer to getting many nights of good sleep. Oventus has been granted clearance from the FDA for the O2Vent T, their second generation 3D printed device created by Dr. Chris Hart, a dentist in Brisbane who worked with the company to provide a solution for patients who cannot be treated effectively with existing treatments such as nasal obstructers and who are CPAP1 intolerant.
“FDA clearance is consequently an important milestone for us as we strive to bring our innovative product portfolio to the global market to benefit those that suffer from OSA or snore excessively,” said chief executive Neil Anderson.
With this US FDA clearance, now they will be able to bring their sleep apnea device to the US and begin launching it through multiple clinical locations. Beta sites are currently being set up, with a formal launch set for October, said the company in their most recent press release.
The first generation O2Vent was already shown in clinical testing to successfully treat sleep apnea through either eliminating snoring or reducing it significantly. This was demonstrated in all patients in the study, according to Oventus, also stating that the positive results included those people who had nasal obstructions and mainly breathed through their mouths, including when they were asleep.
“The recent clinical data strongly supports its superior performance and clearly demonstrates its effectiveness in treating a range of sleep disorders. It also improves oxygen levels for patients,” said Dr. Hart.
“It means a greater number of patients who are CPAP intolerant or mild to moderate sufferers of sleep apnea now have an alternative treatment option available.”
According to the US National Institutes of Health there are approximately 12-18 million adults living just in the US alone with sleep apnea, and a stunning 80 percent are not receiving effective care, if any at all. This new device is more than just a great example of what new technology can produce, it truly may be the catalyst for allowing a great many to live longer, happier, well-rested lives.
The patent is registered in Australia currently, and the product has been on the market there since July, with another patent pending overseas. The O2Vent T is already is registered with the Therapeutic Goods Administration (TGA) in Australia, which the company expects to help pave the way into key European and Asian markets as well. Overall, the company is expecting the titratable version, the O2Vent T, to have similar or even superior results as the jaw position can be more optimized through the titration mechanism.
Find out more at Oventus Medical. The device, previously called ClearWay, is presented in the video below. Discuss further in the 3D Printed Sleep Apnea Device forum over at 3DPB.com.
https://youtu.be/oGh46rHV7ow
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Divergent & GA-ASI: How Connecting at AMUG Led to an Important Industry Partnership
As I mentioned in my AMUG 2026 overview, I had a lot of delays traveling to Reno, Nevada to attend the conference. So unfortunately, I missed “From Hypercars to Defense...
BLT Touts 100,000 Copper Parts Made
With the significant volume of copper used in electronics, semiconductors, aerospace, defense, and beyond, copper additive has had immense promise from the early days of copper 3D printing at Beamit...
TCT Asia 2026: Empowering Asia · Connecting the World
TCT Asia, now in its 12th year, is more than just a 3D printing exhibition — it embodies its original concept: Time Compression Technology, a vision of making the entire...
Pete Pharma Deal with Atrium24 Signals Path for Broader 3D Printed Drug Commercialization
While I would guess we’re still probably at least several years away from 3D printed drugs being a commonplace reality, the growing number of companies with proprietary additive manufacturing (AM)...