Introducing guidel!ne, a High-Temp, FDA Approved PETG Material for Medical 3D Printing From taulman3D
 Choosing the right material is hard enough for the 3D printing hobbyist – who hasn’t chosen a brand of filament that has left them with a useless lump instead of a viable print? It’s maddening, but for certain industrial applications, the wrong material can go far beyond frustrating. The medical industry, in particular, is subject to extremely strict standards when it comes to the materials that can be used for surgical tools or implants, for obvious reasons. Thus, companies like taulman3D, which formulates high-quality filament targeted towards very specific applications, are in high demand.
Choosing the right material is hard enough for the 3D printing hobbyist – who hasn’t chosen a brand of filament that has left them with a useless lump instead of a viable print? It’s maddening, but for certain industrial applications, the wrong material can go far beyond frustrating. The medical industry, in particular, is subject to extremely strict standards when it comes to the materials that can be used for surgical tools or implants, for obvious reasons. Thus, companies like taulman3D, which formulates high-quality filament targeted towards very specific applications, are in high demand.
According to the Missouri company, the medical 3D printing industry has been clamoring for a material that meets ISO 10993 standards for biocompatible medical devices, and taulman3D has now delivered with a new PETG-based filament called guidel!ine. The high-strength, high-temperature material is already registered with the FDA, and users will be provided with plenty of information in sync with the agency’s “Technical Considerations for Additive Manufactured Devices Draft Guidance for Industry and Food and Drug Administration Staff: Section C: Material Controls.”
A device printed with guidel!ne isn’t automatically FDA/CE certified – the certification process is a lot more complicated than that – but it significantly speeds things along to be able to present a part made with an already tested and approved raw material.
“guidel!ne is sold as a raw material modified in form only into 3d printing line and most important, extruded in a manner that keeps contaminates out of the material during the cooling process, where contaminates and bacteria could be embedded in the soft extrusion,” states taulman3D. “In addition, pH and ORP levels are monitored and adjusted to maintain acceptable levels.”
 In addition, the company is providing plenty of support in terms of tracking and and verifiying the material once it’s been shipped. Each one-pound spool of the filament, which is available in 2.85 and 1.75 mm diameters, is stamped with a QR code embedded with a “traceability and batch key,” and consumers will be able to register each spool with taulman3D. Each vacuum-sealed spool ships in its own box and is labeled with an identification number, and – like taulman3D’s other materials – the filament is completely clear which allows for easy examination and evaluation of printed parts.
In addition, the company is providing plenty of support in terms of tracking and and verifiying the material once it’s been shipped. Each one-pound spool of the filament, which is available in 2.85 and 1.75 mm diameters, is stamped with a QR code embedded with a “traceability and batch key,” and consumers will be able to register each spool with taulman3D. Each vacuum-sealed spool ships in its own box and is labeled with an identification number, and – like taulman3D’s other materials – the filament is completely clear which allows for easy examination and evaluation of printed parts.
In terms of performance, guidel!ine has excellent thermal properties and is resistant to distortion, allowing the printing of small, finely detailed parts, including watertight hollow tubes, at high temperatures without warpage. It needs only a small amount of retraction and also serves as a good support material for medical grade nylon prints – it has just enough adhesion to provide support and still be easy to remove.
Here are the fine details:
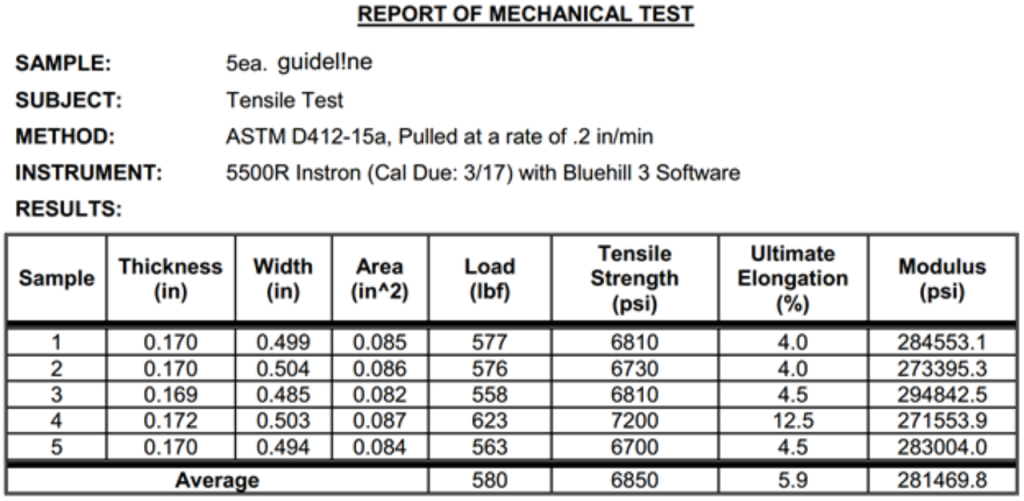
- Tensile strength: 6,850PSI – stronger than taulman3D’s strongest TECH-G material
- Elongation: 5.9, modulus 281,469PSI
- Print temperature: 250°C
- Heat distortion temperature: 70°C
- Softening temperature: 100°C
guidel!ine is now available for purchase from taulman3D’s website. Discuss further in the guidel!ine 3D Printing forum over at 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
RAPID 2026 in Pictures: The Coolest & Craziest Things on the Show Floor
Last week, North America’s premier 3D printing trade show, RAPID+TCT 2026, came to Boston. I spent two days trekking the show floor, trying to see as much as I could,...
ExOne Cuts Costs for U.S. Customers as Printhead Production Moves to Detroit
ExOne Global Holdings, created through the 2025 integration of ExOne and voxeljet, is making changes across its U.S. operations. These include starting printhead manufacturing in the Detroit area and lowering...
Euler Viewer for Metal LPBF 3D Printing Released
Icelandic software startup Euler has released Euler Viewer, a real time build viewer for metal LPBF. The product does not need to be installed, and doesn’t require hardware to be...
3D Printing News Briefs, April 11, 2026: Energy Targets, DoW Contracts, Nike Air Max, & More
We’re starting with 3D printing for energy applications in this weekend’s 3D Printing News Briefs, and then moving on to military and defense 3D printing. Finally, Nike Sportswear is focusing...










































