3D Printed Genetic Models May Help Find Future Cancer Treatments
 There is so much to know about the complexity of life that has already been discovered and yet there is an even greater amount that still remains to be explained. One particular topic that holds a great deal of fascination for researchers involves DNA. From the moment, in 1869, that such a thing as DNA was found to exist through the discovery of its double helix by Watson and Crick in 1953, the substance has tantalized with the promise it holds for unlocking the secrets of life. And one tool that is being used to assist in the study of the minute inner workings of DNA is 3D printing.
There is so much to know about the complexity of life that has already been discovered and yet there is an even greater amount that still remains to be explained. One particular topic that holds a great deal of fascination for researchers involves DNA. From the moment, in 1869, that such a thing as DNA was found to exist through the discovery of its double helix by Watson and Crick in 1953, the substance has tantalized with the promise it holds for unlocking the secrets of life. And one tool that is being used to assist in the study of the minute inner workings of DNA is 3D printing.
Today, researchers from Van Andel Research Institute, Brookhaven National Laboratory, and Stony Brook University in the US, and Imperial College London in the UK have released a paper that details the latest small but profound advancement in understanding the mechanisms by which DNA copies itself. It is well documented that in order for DNA to engage in this near magical act of self replication, it must expose the highly important information that it holds in the seam of its double helix. It does this by ‘unzipping’ itself and then, when the copy is complete, re-zipping itself to continue to protect those important contents.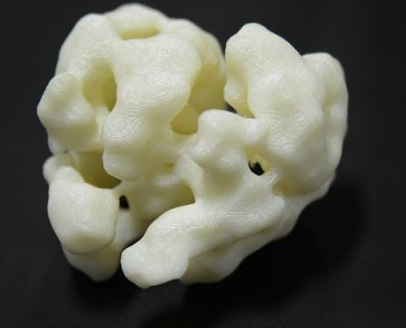
What is not as fully understood is the process by which this assembly-de-assembly takes place. The machinery which allows DNA to operate and survive is highly complex, involving hundreds of components that have to work together just exactly so, or the whole process breaks down. One of those components, rather unpoetically named Cdc6, is a key protein that these researchers theorized was involved in this unzip/zip process, the question was: how?
There is a vital component in this process, an enzyme called DNA helicase, and if we continue to think about this in terms of a zipper, the helicase acts as the clasp at the top of the zipper that allows it to be opened or closed. Prior to this latest research, it was believed that the protein Cdc6 played the part of placing that clasp at the top of the strand. In order to determine whether or not this is the case, the researchers worked to inhibit the Cdc6 and studied the impact that had on the ability of the strand to come undone. Had this protein been responsible for attaching the clasp, its elimination would have meant that the zipper was stuck, unable to open. However, what the researchers found was that the double helix continued to unzip just as before but never made it farther down the path towards replication.
To make a long, and very difficult, paper short: Cdc6 seems to be some sort of enabling protein, something that gives the final go ahead before the copying begins. At this point, it is still somewhat uncertain, but Dr. Christian Speck, head of the DNA Replication Group at Imperial College’s Clinical Sciences Center, explained in laymen’s terms what they think they have found:
“Imagine that you leave a spanner [wrench] in an engine, or don’t remove some of the tools that you used to assemble it. The engine will become jammed and stop working. So, Cdc6 motor activity makes sure there’s no spanner in the works, and keeps the production line going. It’s a sort of quality control protein.”
If you are waiting for the ‘so what’ moment for the general public, here it is:
Abnormal cell growth is extremely problematic because of those cells’ ability to self replicate. Cancer treatments typically focus on destroying the DNA of these abnormal cells. However, because of the nature of those methods, healthy cells can also have their DNA destroyed. If scientists were able to develop a way to simply turn off the part of the replication mechanism that allows the cell to make its copy, then that opens up a whole new world of possibilities for cancer treatment.
In order to even begin to think about the application of this understanding to the treatment of individuals suffering from cancer, a great deal more needs to be understood about not just Cdc6 but all of the different components that come together to build the DNA machine. In Speck’s lab, the team is not only able to examine the delicate structures through electron microscopes, but is also using those images to create and 3D print out large scale 3D models for study. The ability to see these models and handle them is a key aspect to continuing to develop our understanding. Co-author Huilin Li, a biologist at Stony Brook, explained how being able to see the models as a 3D printed version assisted in the research process:
“The genesis of the DNA unwinding machinery is wonderfully complex and surprising. Seeing this helicase enzyme prepare to surround and unwind the DNA at the molecular level helps us understand the most fundamental process of life and how that process might go wrong.”
Via the 3D printed models researchers found that if they blocked the Cdc6 protein from joining the “machine”, the “machine” would then jam, meaning that the DNA copying process would be brought to a halt. 3D printing’s ability to quickly produce physical models of data gathered into 3D images by an electron microscope means that the researchers can have ready access to models as needed and can quickly make adjustments without great expense or long periods of waiting. Now that they have answered one question, the team is ready to take on the next, and even though are asking about microscopic elements, the answers can have enormous consequences.
Let’s hear your thoughts on this research in the 3D Printing and Cancer Treatment forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: Stratasys Bets on Defense and Drones as Printer Sales Slow
Stratasys (Nasdaq: SSYS) started 2026 with lower revenue and a larger loss as customers continued to slow down spending on new 3D printers. Still, the company pointed to stable recurring...
3D Printing Financials: Xometry Surges After Record Quarter and Siemens Deal
Shares of Xometry (Nasdaq: XMTR) surged on Thursday, May 7, after the company reported record first-quarter 2026 results and announced a major partnership with Siemens. The stock climbed as much...
3D Printing Should Benefit Greatly as EU Releases €6 Billion for Ukrainian Drones
Drones have changed everything. Case in point: with the aim of preventing Iran from developing a nuclear weapon, the US and Israel initiate airstrikes on the Gulf nation. A couple...
6K Energy Secures 7-Year Agreement to Supply CRG Defense with Battery Materials
Last year, the US Federal Communications Commission (FCC) announced a ban on certain components sourced from foreign suppliers, including Chinese firms, used in unmanned aerial systems (UAS). Meanwhile, the FY...