 If you were to read our site on a consistent basis, you would begin to think that China was leading the way in 3D printed medical breakthroughs. This is simply because laws in the US and Europe tend to aim at protecting patient confidentiality, more so than in other countries such as China. Fact is though, that each and every month there are plenty of surgeries that take place in the United States and much of Europe, thanks in part to 3D printing technology.
If you were to read our site on a consistent basis, you would begin to think that China was leading the way in 3D printed medical breakthroughs. This is simply because laws in the US and Europe tend to aim at protecting patient confidentiality, more so than in other countries such as China. Fact is though, that each and every month there are plenty of surgeries that take place in the United States and much of Europe, thanks in part to 3D printing technology.
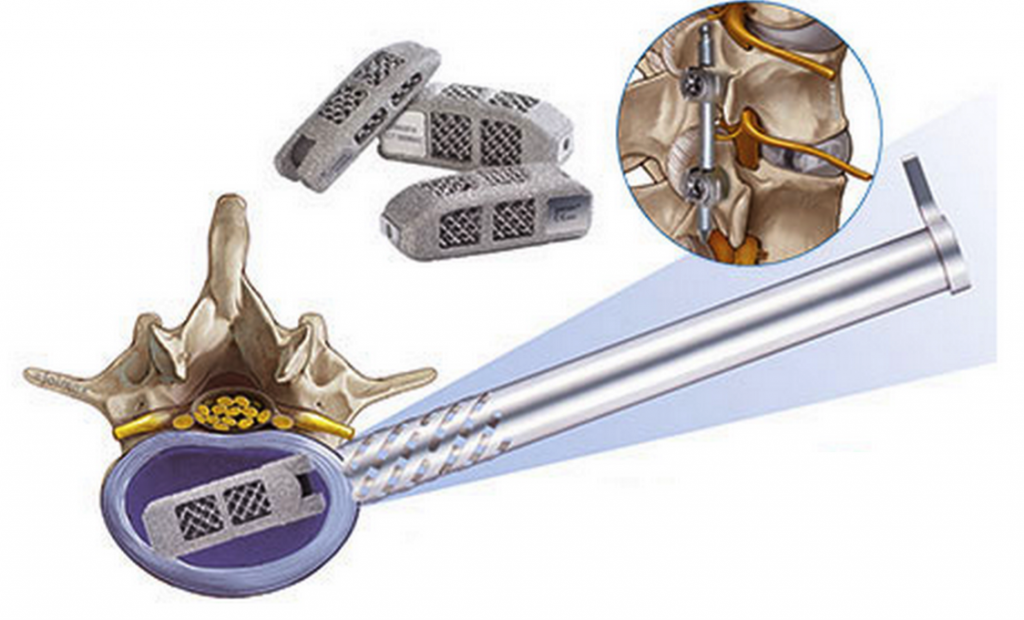
Today, a major announcement was made on behalf of a German company called joimax®. joimax® is the developer behind various minimally invasive methods of endoscopic spinal surgeries. Today they have officially announced that they have received 510(k) clearance from the U.S. Food and Drug Administration (FDA) in order to market their Endoscopic Lumbar Interbody Fusion (EndoLIF® On-Cage implant) in the U.S.
The EndoLIF® On-Cage is a 3D printed titanium implant which is printed using EBM (Electron Beam Melting) technology out of a titanium alloy called Ti6Al4V. It is printed in such a way that the cage features a very porous surface with diamond cell structure. This provides a means for cells to proliferate, and bone to grow onto the metal. Also, as you can see in the photos provided, the EndoLIF features two holes which can be filled with autogenous bone. This allows for the creation of a straight column for fusion to take place.
“Titanium and titanium alloys are well known for their excellent biocompatibility and affinity to the surrounding bone,” the company explains. “Compared to other implant materials, such as recent studies show2, especially the in-corporation behavior of Ti6Al4V ELI alloy (ELI = Extra Low Interstitials) is highly osteointegrative”
 The special design of the implant allows for X-rays and CT scans to verify the growth of bone after it is implanted into a patient.
The special design of the implant allows for X-rays and CT scans to verify the growth of bone after it is implanted into a patient.
“With the EndoLIF program, joimax offers a complete endoscopic-assisted solution for spinal stabilization and fusion. In the future, we will be able to treat patients with even more gentle techniques,” explained Wolfgang Ries, CEO and founder of joimax. “Our next development will be an EndoLIF Cage on the basis of our iLESSYS Delta system for posterior lumbar inter-body fusion (PLIF).”
The EndoLIF allows surgeons to take an “inter-muscular” approach to back surgery, in a method similar to a mini transforaminal lumbar inter body fusion (TLIF). This provides for endoscopic assisted fusion.
While we continue to hear of various stories out of China concerning 3D printing in the operating room, two surgeons from Germany (Dr. Ralf Wagner, LIGAMENTA Spine Center, Frankfurt and Dr. Bernd Illerhaus, ONZ, Datteln/Recklinghausen) have used these EndoLIF implants on 200 patients already, and 600 surgeries in all have been performed using the technology in Europe.
“The access is dura and nerve-gentle, preserves the dorsal bony structures and we can avoid scar tissue because of the stepwise tissue dilation,” said Dr. Illerhaus.
It should be interesting to see how well this implant catches on in the United States, now that it has FDA approval. What do you think about this unique approach of conducting back surgery on patients? Discuss in the EndoLIF forum thread on 3DPB.com. Check out the video below, showing the process used by surgeons while implanting these devices.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Nikon SLM Solutions Sells SLM 500 to Primary Weapon Systems to Expand Suppressor Production
Primary Weapons Systems (PWS) is a Boise, Idaho-based manufacturer of suppressors, firearms, and related components. A subsidiary of Vigilant Gear and a sister company to aftermarket Glock slide manufacturer Lone...
3DPOD 261: Tooling and Cooling for AM with Jason Murphy, NXC MFG
Jason Murphy´s NXC MFG (Next Chapter Manufacturing) is not a generalist service; instead, the company specializes in making tooling. Using LPBF and binder jet, the company produces some of the...
HP and Firestorm Labs Form Partnership to Use Multi Jet Fusion 3D Printers in Deployable Factories
HP Inc., maker of a range of additive manufacturing (AM) solutions including the Multi Jet Fusion (MJF) ecosystem, has announced a partnership with Firestorm Labs, a developer of containerized, deployable...
3D Printing News Briefs, July 2, 2025: Copper Alloys, Defense Manufacturing, & More
We’re starting off with metals in today’s 3D Printing News Briefs, as Farsoon has unveiled a large-scale AM solution for copper alloys, and Meltio used its wire-laser metal solution to...

































