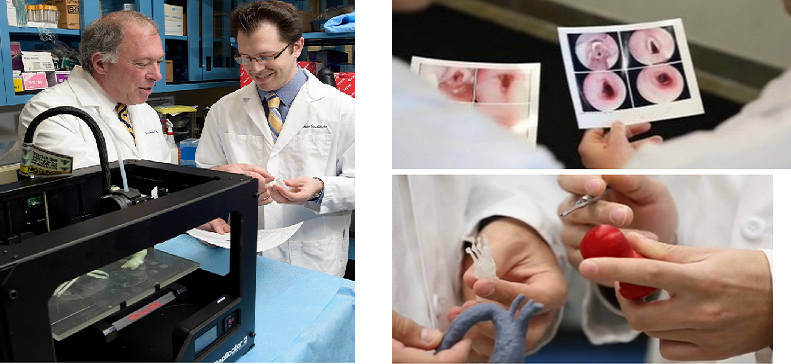
Doctors at the Feinstein Institute for Medical Research, part of the North Shore-LIJ Health System in New York, say 3D printing can be used to create a biodegradable tracheal segment containing a patient’s own cells to aid them in complex reconstruction surgery.
Also known as the “windpipe,” the trachea is the tube that connects the upper respiratory tract to the lungs.
The traditional treatments for tracheal diseases stenosis and malacia now involve removal of the affected tracheal segment.
“Three-dimensional printing and tissue engineering has the potential for creation of a custom-designed tracheal replacement prosthesis in the lab so that the affected tracheal segment can be ‘swapped out’ instead of removed,” says Todd Goldstein of the Feinstein Institute for Medical Research. “Our results show that three-dimensional printing can be combined with tissue engineering to effectively produce a partial tracheal replacement graft in vitro. Our data demonstrate that the cartilage cells seeded on the graft retain their biological capability and were able to proliferate at the same rate as native cells.”
The team made cartilage from a mixture of cells called chondrocytes, used nutrients to feed them, and employed collagen to bind the materials together. But the key was shaping the resulting 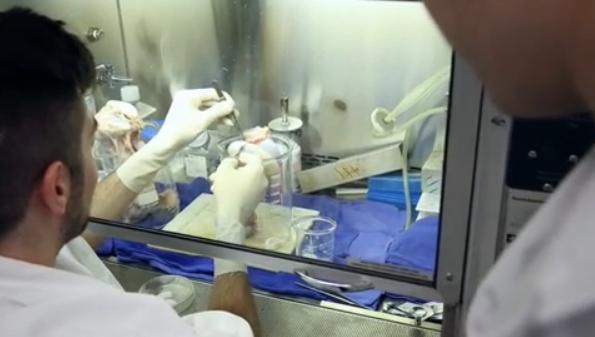 cartilage into a windpipe by constructing a scaffold they cover with the chondrocytes and collagen.
cartilage into a windpipe by constructing a scaffold they cover with the chondrocytes and collagen.
“Making a windpipe or trachea is uncharted territory,” Goldstein says. “It has to be rigid enough to withstand coughs, sneezes and other shifts in pressure, yet flexible enough to allow the neck to move freely. With 3D printing, we were able to construct 3D printed scaffolding that the surgeons could immediately examine and then we could work together in real time to modify the designs. MakerBot was extremely helpful and consulted on optimizing our design files so they would print better and provided advice on how to modify the MakerBot Replicator 2X Experimental 3D Printer to print with PLA and the biomaterial.”
Goldstein and his colleagues used their custom-designed 3D printer to create a series of tracheal segments from living cells which can be tailored to the precise anatomy of each patient.
The three types of printed segments — empty segments, control segments without cells, and segments that had been combined with living cells — were bio-printed and tested for viability, cell growth and division, and gene expression. The cells survived the printing process to ultimately produce material with the cellular properties they expected of healthy tracheal cartilage.
“We think the next phase will be integrating three-dimensional printing and tissue engineering to produce customized biological replacement parts,” said Dr. Goldstein. “While further development is necessary before a clinical trial would be viable, our results show that 3D printing technology is a feasible alternative to traditional treatments.”
 Goldstein has been working with a team of surgeons at the North Shore-LIJ Health System for the past year on the tissue engineering project, and he says it could be used for tracheal repair and replacement.
Goldstein has been working with a team of surgeons at the North Shore-LIJ Health System for the past year on the tissue engineering project, and he says it could be used for tracheal repair and replacement.
Goldstein and Daniel Grande, PhD and director of the Orthopedic Research Laboratory at the Feinstein Institute, worked with Lee Smith, MD, the chief of pediatric otolaryngology at Cohen Children’s Medical Center, and David Zeltsman, MD, the chief of thoracic surgery at Long Island Jewish Medical Center, for the last year on the project.
“The ability to prototype, examine, touch, feel and then redesign within minutes, within hours, allows for the creation of this type of technology. If we had to send out these designs to a commercial printer far away and get the designs back several weeks later, we’d never be where we are today,” Dr. Smith says.
The work was done using standard PLA materials, and that material provided an additional advantage. Testing done by Goldstein showed that heat generated by the extruder head sterilized the PLA as it was printed.
“PLA is… used in all kinds of surgical implant devices,” says Dr. Smith.
During the process, the “bio-ink” the researchers use stays at room temperature and cures into a gel on the heated build plate as two-inch-long sections of windpipe are printed in less than two hours. As the bio-ink adheres to the scaffolding, it’s then placed in a “bioreactor,” a sort of oven.
The results of the work were presented before the Society of Thoracic Surgeons.
While the method will require approval from the Food and Drug Administration (FDA), the doctors say that, if no approved treatment exists for an ailment, the FDA can grant what’s called a compassionate therapy exception which would allow patients to sign off on the use of experimental approaches.
“It’s completely changed the trajectory of my academic career,” Goldstein said.
Goldstein is now the Feinstein Institute’s 3D printing specialist, printing models of organs for pre-operative planning and tools aimed at improving the lab there.
It seems that 3D printing is now a go-to method in the toolbox for doctors and surgeons working on complex reconstructions and treatments. Do you know of any other novel work being done in medicine with 3D printing? Let us know about it in the 3D Printed Trachea forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
ExOne + voxeljet Are Trying to Do the One Thing Customers Need Right Now: Keep Machines Running, and Rebuild Confidence
For years, ExOne and voxeljet were two of the best-known names in binder jet 3D printing, especially in sand printing for foundries. But in recent years, ExOne “got buried” inside...
BigRep Launches ONE.5X 3D Printer, Announces New Massive Dimension Partnership at RAPID + TCT 2026
As the whole world is starting to realize, the Hormuz supply chain fallout is only just beginning to filter into the global economy, and the rising cost of plastics should...
Orano Federal Services & UNC Charlotte Show How AM Could Cut Costs in Nuclear Energy Resurgence
Outside of the defense sector, few industries have been impacted by Russia’s ongoing occupation of Ukraine more than nuclear energy. The same appears to already be happening in response to...
EOS to Spotlight AI, Robotics, and Industrial Tooling at Hannover Messe
The US-Israel war on Iran is already catalyzing the sorts of major shifts to global supply chains that will effectively amount to permanent economic changes. In this context, the nations...