6K Additive and RICOH Announce Advances in Medical 3D Printing at Rapid + TCT
The RAPID + TCT event has finished its 2023 run, but not before 6K Additive and RICOH made significant announcements related to medical 3D printing. 6K Additive announced its partnership with Surgical Metal Recycling to create a circular, sustainable supply chain for medical implants, utilizing advanced metal powder production technology. Meanwhile, RICOH announced an expanded FDA (U.S. Food and Drug Administration) 510(k) clearance for its healthcare 3D printing technology, enabling more comprehensive 3D models of soft tissue anatomy.
These announcements demonstrate the innovative solutions being developed in the field of medical 3D printing. The partnership between 6K Additive and Surgical Metal Recycling addresses critical sustainability challenges and ensures a reliable supply chain for medical implants. Similarly, the expanded FDA clearance for Ricoh’s 3D printing technology for healthcare highlights their commitment to improving patient outcomes.
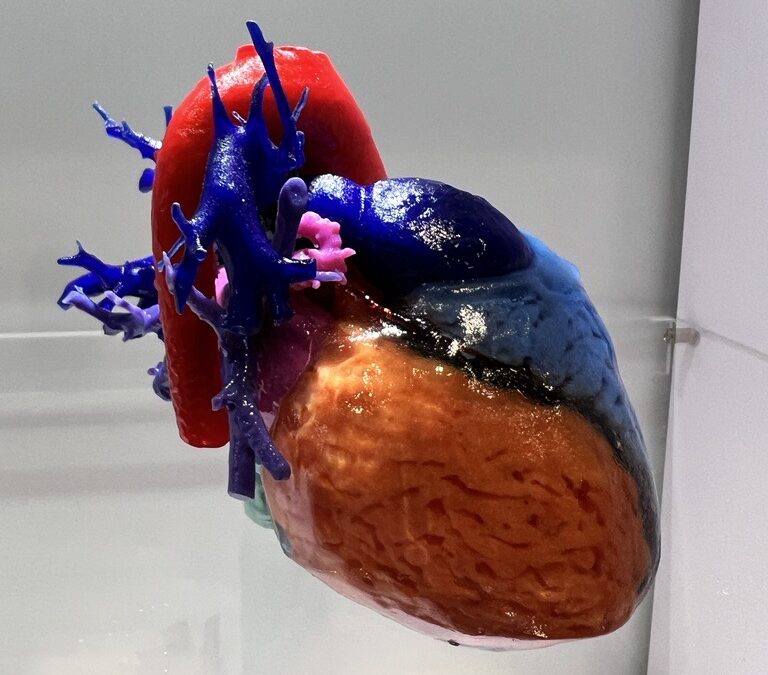 RICOH 3D for Healthcare can 3D print anatomical models of soft tissue. Image courtesy of RICOH.
RICOH 3D for Healthcare can 3D print anatomical models of soft tissue. Image courtesy of RICOH.3D Care
During the last day of RAPID + TCT, 6K Additive, an advanced metal powder production technology developer, announced the deal with Australian firm Surgical Metal Recycling to explore a shared mission to transform the metals supply chain in Australia. The move is expected to directly impact healthcare by revolutionizing how surgical implants are treated at the end of their useful life. Thanks to 6K’s UniMelt process, end-of-life and out-of-spec parts can be profitably reused with a vastly reduced environmental footprint.
Hip, knee, and shoulder joint replacement surgeries, along with the increasing use of metal nails, screws, and plates as part of surgical interventions, have created a significant deposit of valuable metals and alloys within the human population. When implants are removed for replacement or postmortem, there are limited options for recycling and reusing the materials. For example, the implant may be cleaned and sterilized for future use in another patient. However, reusing implants carries some risks, such as the possibility of infection or other complications. Alternatively, the removed implant may be sent to a medical waste management company for safe and environmentally responsible disposal. Depending on the type of implant and the regulations in the region, the implant may need to be treated as hazardous waste.
Another option is recycling the implant. In recent years, there has been an increasing interest in recycling medical implants, particularly those made of metal. As a result, some companies are exploring more sustainable and efficient recycling methods, such as 6K Additive’s partnership with Surgical Metal Recycling, which uses advanced metal powder production technology to transform used medical implants into high-quality metal powders for 3D printing applications.
 A hip cup 3D printed with used and out-of-spec implant powder. Image courtesy of 6K Additive.
A hip cup 3D printed with used and out-of-spec implant powder. Image courtesy of 6K Additive.Thanks to the new agreement, the two organizations will take used and out-of-spec implants, swarf, and used metal additive manufacturing (AM) powder supplied by SMR and reprocess the material through 6K Additive’s UniMelt production-scale microwave plasma platform initially in the U.S. and later moving to Europe. The resulting premium powders can then create new parts through AM. Still, the partnership’s ultimate goal is to generate newly certified implants from existing parts through a sustainable and circular supply chain.
Frank Roberts, President of 6K Additive, explained, “There is a growing population that requires medical titanium implants for knees, spine, and hips; this agreement creates a path to recycle these parts and enable new implant production with sustainably sourced feedstock.”
Although the collaboration initially focuses on titanium, it will eventually expand to include cobalt chrome. According to 6K, their UniMelt process can remove oxygen from titanium powder and improve the material grade. The process also produces over 90% yield of the desired particle size distribution, higher than typical yields of 25% to 35% from other plasma or gas atomization processes. This further reduces cost and environmental impact, according to the company.
Anatomical crafting
RICOH’s 3D for Healthcare, a 3D medical manufacturing center for the development, design, and production of 3D printed anatomic models, received an expanded FDA 510(k) clearance from the FDA for 3D anatomic modeling of new areas, including cardiovascular, neurological, gastrointestinal, genitourinary, and breast applications. The expanded clearance empowers Ricoh to support more surgical specialties and patient diagnoses to print bony and soft tissue models using Stratasys 3D printing technology and materials.
 RICOH’s 3D printed anatomical model of soft tissue. Image courtesy of RICOH.
RICOH’s 3D printed anatomical model of soft tissue. Image courtesy of RICOH.Commenting on the company’s latest achievement, RICOH Managing Director of AM North America said this is “an exciting milestone” as the business continues offering “democratized access to patient-specific 3D printed models in healthcare.”
The manufacturer’s end-to-end workflow solution integrates with Merge Universal Viewer (formerly IBM iConnect® Access), an enterprise imaging solution from Merative (formerly IBM Watson Health) used in many hospitals across the United States. This allows providers to request 3D printed anatomic models using RICOH’s centralized print-and-ship solution for same-week delivery or produce the models at a RICOH-managed, point-of-care production facility onsite at the healthcare facility for next-day delivery.
Relying on segmented 3D print files created from medical images in FDA-cleared applications, RICOH’s patient-specific models are used for diagnostic purposes in various medical fields.
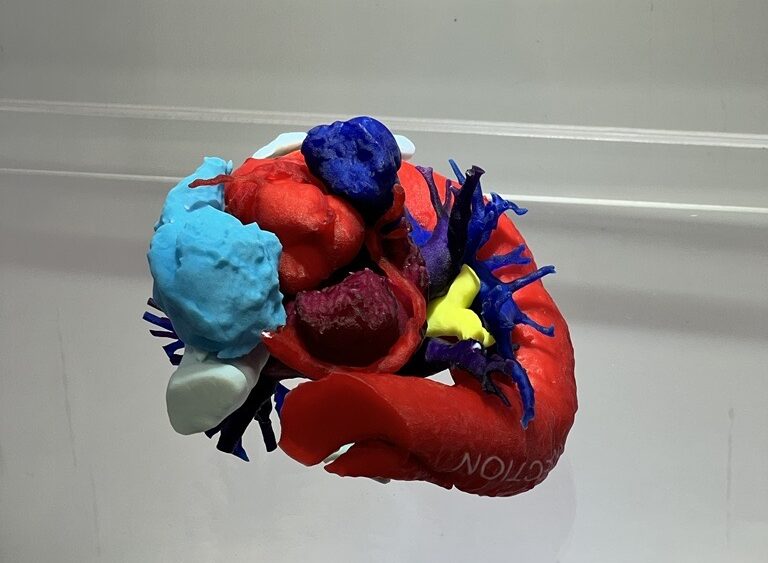 RICOH’s 3D printed anatomical model of soft tissue. Image courtesy of RICOH.
RICOH’s 3D printed anatomical model of soft tissue. Image courtesy of RICOH.Overall, the RAPID+TCT 2023 event provided a valuable platform for companies like 6K Additive and RICOH to showcase their latest innovations in the rapidly evolving field of medical 3D printing.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing News Briefs, April 30, 2026: Support-Free Titanium, Drug Delivery, & More
In today’s 3D Printing News Briefs, we’ll start with Makelab’s new website, and move on to commercialization of support-free metal 3D printing in South Korea. We’ll end with drug delivery...
Post-Printing at RAPID 2026: PostProcess Technologies & Mass Finishing
The whole 3D printing workflow was on display in Boston at RAPID+TCT last week, from design software and 3D printing hardware all the way to post-processing and finishing solutions. For...
RAPID 2026 in Pictures: The Coolest & Craziest Things on the Show Floor
Last week, North America’s premier 3D printing trade show, RAPID+TCT 2026, came to Boston. I spent two days trekking the show floor, trying to see as much as I could,...
RAPID Roundup 2026: New Machines and Market Moves
RAPID + TCT 2026 wrapped up yesterday, but the show floor proved there’s a lot happening across the additive manufacturing (AM) space, especially when it comes to new hardware and...













































