$23M Funding Round to Expand 3D Printed Surgical Solutions from restor3d
A Research Triangle Park-based medical device company that is looking to reconstruct and repair the human body with 3D printing technology has raised $23 million in its fifth venture round. restor3d, a Duke University spinoff that designs and manufactures biomedical implants using its 3D printing technology platform, has now raised a total of $42 million in funding from over 100 investors.
Known for giving surgeons the tools to reconstruct and repair portions of the human body with patient-specific 3D printed implants, restor3d merges 3D printing, biomaterials, biomechanics, and artificial intelligence. It specializes in custom implants used for trauma and oncology cases, as well as off-the-shelf, 3D-printed implants that have been cleared by the Food and Drug Administration (FDA) for foot and ankle, as well as spinal applications.
Although the identity of the backers for this investment round was not revealed, restor3d did say it would use the funds to expand the delivery of 3D printed personalized surgical solutions across multiple musculoskeletal specialties, including the upper and lower extremity joints, spine, and trauma from musculoskeletal injuries. Additionally, the startup will develop machine learning software tools to assist engineers with patient-specific implant design; engage in fundamental research for preclinical and clinical studies and expand the commercial team to sustain its market traction.
To support the company’s growth, in early 2023, restor3d will move into a new facility less than an hour away from its current Durham, North Carolina location. The new site at the top high technology Research Triangle Park will expand its capabilities for patient-specific digital design, in-house manufacturing of implants and instruments, as well as surgeon training and education labs. Sharing the location with other 3D printing companies in the area, restor3d will be immersed in a unique community, with the potential for upcoming collaborations and partnerships. Other up-and-coming startups at the park include 3D software company Geomagic – now part of 3D Systems –, and 3D printing service provider Plastibot.
Engineering professor at Duke University Pratt School of Engineering and restor3d Co-Founder and Chief Technology Officer (CTO) Ken Gall said he is “looking forward to entering our next phase of development” and appreciates the investors for “supporting our growth and the hard work of all our employees, surgeons, and distributors in helping to advance patient care.”
Implants for the future of surgery
Founded in 2017, restor3d came out of stealth mode after 2018, and its products and technology are based on Gall’s scientific research and development in 3D metal printing at Duke. restor3d has since used laser powder bed fusion of medical-grade polymers and metals to create proprietary implants, like its osteotomy wedges and cervical fusion cages. It also aims to update the implant industry by luring surgeons to deliver 3D printed personalized surgical solutions across multiple musculoskeletal specialties.
During a recent presentation, Gall said orthopedic reconstructions have been done through a “brute force solution,” even though it has always been a patient-specific need. Instead, restor3d hopes pre-surgical planning and 3D printed patient-specific instruments and implants become part of all orthopedic surgeries in the future.
Thinking primarily about the patient, Gall is a firm believer in leveraging 3D printing technology for surgical procedures, even to avoid amputation. A few years ago, Gall was charged with designing and printing a custom titanium implant for a young patient who had suffered critical damage to a bone in her lower leg during a vehicle accident.
“There was a lot of bone loss, but it was definitely worth salvaging her leg even though many centers would have probably amputated,” explained Duke orthopedic surgeon Samuel Adams. “I immediately thought of implanting a 3D printed cage to replace the lost bone.”
Working with Adams, Gall’s group used the patient’s CT scans to inform the fabrication and design of a scaffold to support the patient’s weight while healing and growing new bone around the titanium structure. The result was a large, four-inch piece of porous and solid titanium that would replace the patient’s damaged distal tibia.
The challenge with leg or ankle implants, says Gall, is that they must be designed to be weight-bearing during the healing process, but must also promote bone growth; other complex factors include focusing careful attention not only on designing the anatomical external shape of the implant but considering how each patient’s bones might grow based on their anatomy. Hopefully, what Gall is suggesting will become the bread and butter of the implant industry in the future.
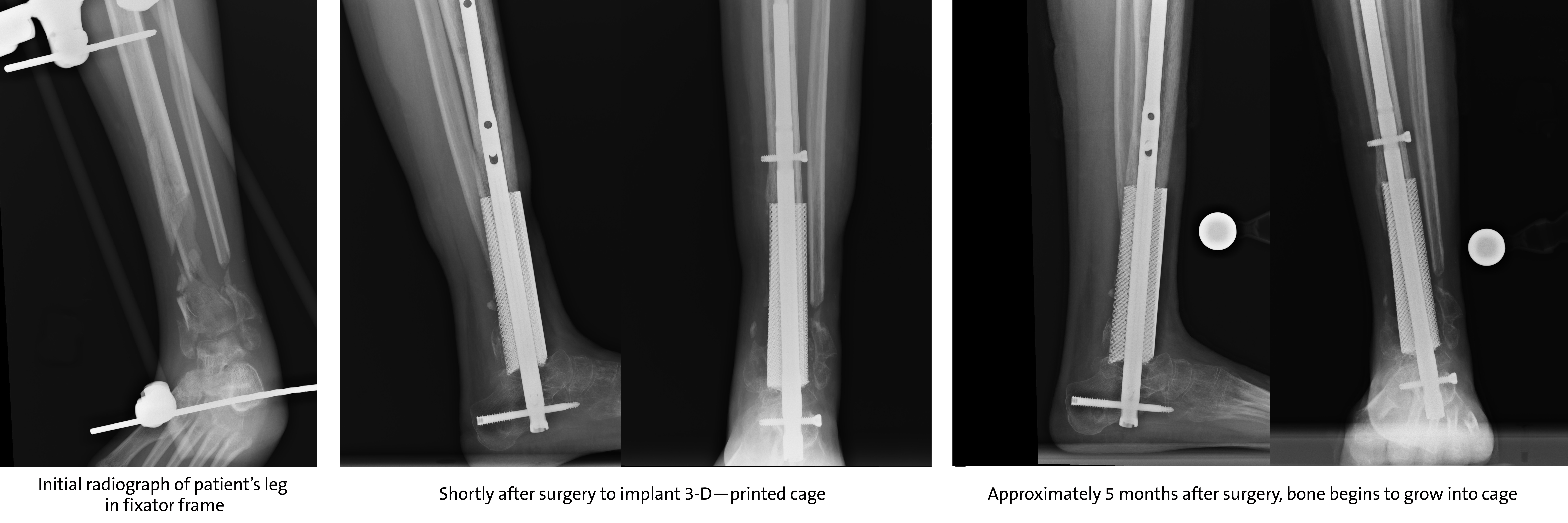
restor3d’s 3D printed implant replaces distal tibia bone after a serious accident. Image courtesy of DukeHealth.
Nearing 100 employees, restor3d has actively patented six products, including custom surgical devices and implants that promote osseointegration (that is, bone ingrowth into a metal implant). The startup has been 3D printing implants with biomaterials such as titanium and cobalt chrome as well as biocompatible polymers and has worked closely with surgeons in the US to aid patients with severe orthopedic needs.
For example, with Dr. Amiethab Aiyer, an orthopedic surgeon at the University of Miami (UM) in Florida, restor3d helped create a total talus replacement using a prosthetic metal bone 3D printed out of cobalt chrome, for a patient that suffers from a debilitating sickle cell disease, a genetic disorder that blocks blood flow and, in her case, caused avascular necrosis in her talus bones. Described by the institution as a “Terminator-style, 21st Century approach,” the procedure, which took place in 2019, was successful and enough to put the Miller School of Medicine at the forefront of using 3D printing technology to create orthopedic solutions.
In one of its most recent moves, restor3d announced a merger with Kinos Medical, a leading total ankle replacement company, which helped accelerate restor3d’s expansion into the high-growth extremity arthroplasty markets in the U.S. One of its most well-known products is the proprietary Kinos Axiom Total Ankle System, which is described by the company as the first biomechanically accurate ankle system that mimics a patient’s healthy range of motion while walking.
restor3d has multiple FDA product clearances as well as collaborations and partnerships that make the company one of the most innovative in the sector. With the new funding and its 3D printed implant and disposable instrument model, restor3d will enable simultaneous expansion across these diverse markets to provide innovative solutions for pressing clinical needs.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
HP’s MJF 1200 Targets Entry-Level AM — And Could Shift the Competitive Landscape
HP has launched an entry-level MJF solution, the HP Multi Jet Fusion 1200 3D Printer Solution, which will be available in 2027. What is the 1200 exactly? What does it...
Metal Base & Scrap Labs Usher in an Ultra-Low-Cost Sub-$10,000 Metal LPBF Evolution
Above, an image of a part coming off the ScrapLabs Scrap 1. Dutch firm Metal Base started a low-cost metal LPBF revolution last month. An ultra-low-cost metal laser powder bed...
Most 3D Printing Metals Are Adapted. This One Was Designed with AI
A research team from the University of South China and Purdue University developed a new type of steel designed specifically for 3D printing, using machine learning to guide the process....
Nikon AM Synergy Gets Defense Innovation Unit FORGE Contract
Nikon AM Synergy has received an Other Transition Agreement (OTA) contract from the U.S. Department of War (DoW) Defense Innovation Unit (DIU). The Foundry for Operational Readiness and Global Effects...