ROKIT Healthcare’s bioprinting-based diabetic foot treatment kit registered as a U.S. FDA medical device

ROKIT Healthcare has completed registration of its Dr. INVIVO-based diabetic foot treatment with the U.S. FDA and has successfully completed clinical studies in the U.S.
Since 2019, the company has conducted clinical studies in India, Korea and Turkey, and based on the most recent study results in the U.S., the treatment has shown promising results in all of the study patients with no side effects.
The company reported that the study subjects were patients of chronic diabetic foot ulcer with an average of 64.4 years of age, 15 years of diabetes duration, and obesity in 90% of the patients.
The principal investigator behind the study is Dr. David G. Armstrong, PhD, DPM, who is Professor of Clinical Surgery at University of Southern California and has produced more than 510 peer-reviewed research papers in scholarly medical journals and 90 book chapters with expertise in the treatment and clinical care of the diabetic foot. Dr. Armstrong is past Chair of Scientific Sessions for the American Diabetes Association’s Foot Care Council and the founder and co-chair of the International Diabetic Foot Conference, the largest annual international symposium on the diabetic foot in the world.
“There is no existing method that resolves chronic diabetic foot ulcers by way of true regenerative medicine, especially for those ulcers that failed to close for many months,” said an official of ROKIT Healthcare. “We have seen our bioprinting-based diabetic foot ulcer treatment offer hope for patients who recovered and got back on their feet within just a few weeks after one procedure, making daily life possible again.
“The clinical study results have demonstrated that ROKIT Healthcare’s diabetic foot ulcer treatment kit can effectively regenerate skin of diabetic foot affected areas regardless of various variables such as race, age, and underlying disease. I can take one more step,” he said.
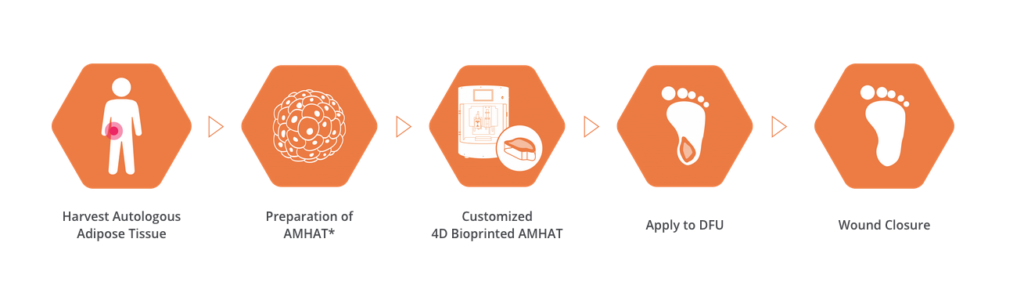
ROKIT Healthcare’s diabetic foot ulcer treatment, which consists of the printer Dr. INVIVO DFU and patient-specific autologous solution extraction kits, uses 3D scanning and AI-based interpretation of patient wounds to create customized skin regeneration patches.
“Given the FDA medical device registration and several successful global studies yielding promising results, we are proud to be leading a meaningful work of saving human lives with cutting-edge regenerative medicine technologies,” said Seok-Hwan You, CEO of ROKIT Healthcare. “I look forward to contributing to the advancement of chronic ulcer treatment methods and significantly improving the quality of life for patients.”
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Nikon SLM Solutions Sells SLM 500 to Primary Weapon Systems to Expand Suppressor Production
Primary Weapons Systems (PWS) is a Boise, Idaho-based manufacturer of suppressors, firearms, and related components. A subsidiary of Vigilant Gear and a sister company to aftermarket Glock slide manufacturer Lone...
3DPOD 261: Tooling and Cooling for AM with Jason Murphy, NXC MFG
Jason Murphy´s NXC MFG (Next Chapter Manufacturing) is not a generalist service; instead, the company specializes in making tooling. Using LPBF and binder jet, the company produces some of the...
HP and Firestorm Labs Form Partnership to Use Multi Jet Fusion 3D Printers in Deployable Factories
HP Inc., maker of a range of additive manufacturing (AM) solutions including the Multi Jet Fusion (MJF) ecosystem, has announced a partnership with Firestorm Labs, a developer of containerized, deployable...
3D Printing News Briefs, July 2, 2025: Copper Alloys, Defense Manufacturing, & More
We’re starting off with metals in today’s 3D Printing News Briefs, as Farsoon has unveiled a large-scale AM solution for copper alloys, and Meltio used its wire-laser metal solution to...