Medical Center Completes Clinical Trials of 3D-Printed Nasal Swabs
Upon conducting a clinical trial, a research team from Beth Israel Deaconess Medical Center (BIDMC), part of Beth Israel Lahey Health, has closed in on four novel prototypes of 3D-printed swabs that can be used for COVID-19 testing. The team’s manuscript can be found in preprint form here.
One of the most interesting innovations that has sprung out of the pandemic response is the invention of new designs and production methods for nasopharyngeal (NP) swabs. The ability to 3D print nasal swabs seems to help tackle a very significant gap in the supply chain at an obviously crucial time, while introducing designs only possible with 3D printing that could advance the art of medical testing.
Since the outbreak of the SARS-CoV-2 virus, we’ve seen several 3D printing companies announce research into the development of 3D printable NP swabs. These swabs could work for any testing that relies on samples obtained by scraping the nose or mouth, but are not relevant for tests that collect saliva or blood in a vial. PCR tests work by detecting specific genetic material within the virus, compared to blood tests that mine samples for antibodies to the virus. Rather than introduce too much redundancy into the situation amid a pandemic, these competitors banded together to form a coalition called PrintedSwabs.org, uniting Formlabs, Carbon, Origin and EnvisionTEC with several medical research institutions to produce swabs for various medical facilities short on supplies.
In mid-March, BIDMC predicted impending swab shortages that could hamper polymerase chain reaction (PCR) testing for COVID-19. To head off the issue, the BIDMC team developed a three-stage protocol for obtaining new swabs. Design considerations for the swabs included:
- A length of six inches
- Sufficient rigidness to reach potentially infected cells at the back of the nasopharynx
- Sufficient flexibility to not damage soft and sensitive tissues
- That the swab be made of materials that don’t interfere chemically with the test
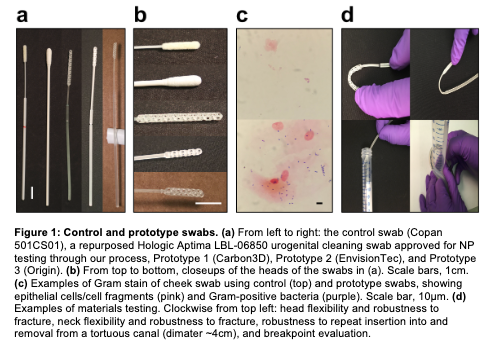
In stage one of the evaluation experts from infectious disease doctors to respiratory therapists provided feedback on over 100 prototypes in less than two weeks. The team obtained 150 novel swab designs made of 45 different materials submitted by 23 companies, laboratories and individuals around the U.S.
Stage two saw the BIDMC team examine the possibility of large-scale production, sterilization and packaging of the swabs, as well as the supply chains necessary to support all of this. This was taken into account “to minimize the risk of future single failure points such as those that contributed to this current swab-shortage crisis,” according to Ramy Arnaout, MD, DPhil, Associate Director of the Clinical Microbiology Laboratories at BIDMC.
The third stage saw initiation of the IRB-approved clinical trial for testing promising prototypes on adult patient-volunteers, including prototypes from Carbon/Resolution Medical, EnvisionTEC, HP and Origin. Involved in the research was New York’s Northwell Health, whose Todd Goldstein was interviewed by 3DPrint.com about the healthcare provider’s work on a 3D-printed BiPAP-ventilator adapter. Goldstein told 3DPrint.com that the project has progressed to the point that the swabs can be produced by the Northwell hospitals themselves:
“Currently, we have six printers going. Each of them is producing 400 swabs a pop running continuously. We have 2,400 swabs printing at a clip and we’re producing them for use in our hospital. Also, with [University of Southern Florida], we have granted access to our intellectual property to any health system that requests it for a minimum of one year. This will allow other hospitals who are experiencing shortages and have the same Formlabs machines can use our validation and print their own swabs for testing.”
Other healthcare providers interested in obtaining the design can fill out a form on Northwell’s website here.
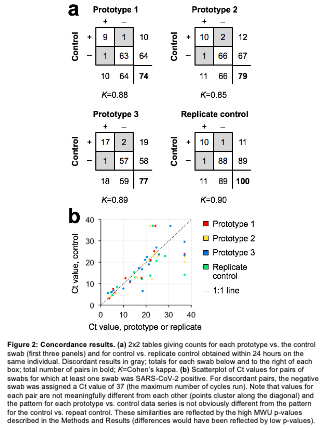
It took just 22 days from the time the group identified the swab shortage to achieve clinical validation of the first new swab. Now, BIDMC has announced that, after testing 3D-printed swabs against standard NP swabs in 230 adults, four designs have shown “excellent concordance with the controls in a clinical trial.”
The clinical trial is ongoing, as the medical team continues to validate the work, but Arnaout said, “We are cautiously optimistic that the availability of swabs will soon no longer be a constraint on widespread COVID-19 testing, thanks to an extraordinarily hard-working, talented, and selfless group, including dozens of people at BIDMC, who came together from all over the country to do this work.”
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing News Briefs, May 30, 2026: RIMPAC 2026, Acquisition, Ceramic Implants, & More
We’re kicking things off with materials news in this weekend’s 3D Printing News Briefs. Then it’s on to a hybrid manufacturing system for a maritime exercise, an expansion of industrial...
The Hidden Cost of Lost Engineering Intent
Recent reporting on the UPS MD-11 engine separation on Flight 2976 has understandably focused on the immediate question: what happened? The investigation will determine that, as it should. Serious engineering...
Scientists Create Stretchy 3D Printed Implants for High Blood Pressure Treatment
Researchers at Pennsylvania State University (Penn State) say they may have found a softer, less invasive way to treat severe high blood pressure. In a new study published in the...
Conexeu Sciences Wishes to Make a Regenerative Breast Matrix Solution (and sell shares)
Conexeu Sciences wants to commercialize a regenerative breast matrix solution. For years, Australian firm BellaSeno and Lattice Medical have been working on similar solutions. In 2018, Lattice came out with...