Costa Rica: Researchers Design 3D Printed Medical Device for Suturing Extremities
Our skin protects us from invading microorganisms and foreign substances, eliminates harmful toxins, helps to regulate our core body temperature, and is in charge of receiving both tactile and thermal stimulation. But, it’s fragile and easily damaged, which can lead to open wounds that get infected. Michelle Orozco-Brenes, José A. Jiménez-Chavarría, and Dagoberto Arias-Aguilar, researchers out of Costa Rica, published a paper, titled “Design of a medical device for superficial suturing upper and lower extremities,” about their work creating a medical suturing device.
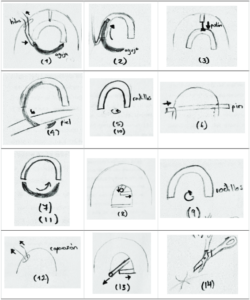
“This work presents the design for a class 2 medical device that meets the basic requirements of the current and known suturing methods in Costa Rica,” the abstract states. “The design process was achieved in three main stages, (i)Research on similar technologies; e.g. The operation principles of a sewing machine, materials used; (ii) The study of types of skin traumas; (iii) General approach toward the suturing device, including device functionality, integration with the human body and manufacturing process. The device model was designed and fabricated using 3D printing technology, this allowed the team to analyze ergonomics, the assembly of the parts and the equipment’s motion. The printed prototype made it possible for potential users to provide feedback on the design and suggestions for improvement.”
Suturing means to connect blood vessels with a specific material, such as thread, when tissue is torn in a way that halts natural healing. You can find many suturing devices on the market around the world, but Costa Rican hospitals don’t typically use them, as they are complex and costly. So the team set out to design a class 2 FDA electronic medical device that could both reduce tissue damage and uniformly, and quickly, suture a wound so an “aesthetically acceptable” scar is left behind.
“The idea for a medical device to suture arose for three main reasons,” the researchers wrote. “First, physicians were noticing poorly sutured wounds that would result in large scars. These in some cases required further procedures like plastic surgery. Also, time consumption, making the search for a device that would make the method faster a necessity. Finally, sutures stitched by hand are sometimes left too loose or too tight, causing bleeding from the wound.”
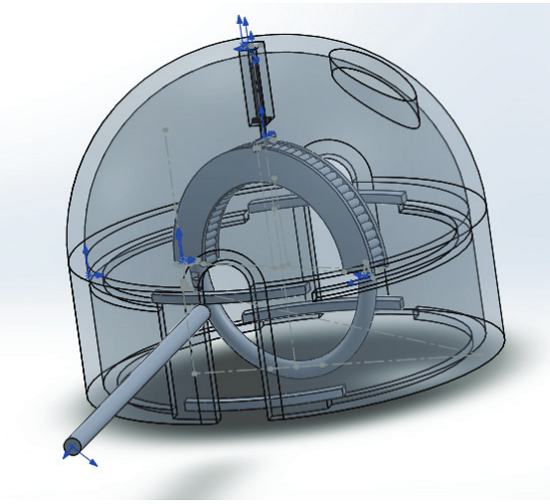
Device specifications were functionality, cost, durability, modularity, and reliability. They used SOLIDWORKS software to create the design for their model, which required three specific functions:
- Stabilize the skin
- Rotate the needle on its axis to join tissue sections
- Initiate and finish with the least possible amount of user interference
“The final design was oriented to have the area and volume of the shell as similar as possible for the needle to rotate 360° without any problem,” the researchers explained.
In order to test out several functionality features, they 3D printed a prototype first, using Polyjet technology to fabricate the piston and and an FDM printer for most of the other parts. Due to its high strength and toughness, corrosion and fatigue resistance, and low friction coefficient, they used the AISI 316L alloy for the prototype.
The suturing device has seven main components. The shell encases the device, while two guides allow the movement of the guide pin, which is used to tie a double knot. Rollers provide the rotational movement that allows for the suturing, while a piston gives the rollers their movement. The final parts are a ½ circle needle with tapered tip, and nylon thread, which has good elasticity for skin retention and closure.
To use the device, the needle is first threaded in its initial position at the top of the shell, and then set in the rollers. The piston lowers the shell, and the needle is rotated 270° to pinch the tissue for suturing. The knot is initiated when the rollers, guided by the holder, turn 45° to the right, and the pin is set in place over the guide. The needle makes a 360° turn on its axis, and the guides turn over the shell and let go of the guide pin, “letting it fall due to gravity over the guides” beneath it to finish the first knot. The first few steps are repeated, and after the final full turn, the user tenses the thread through the top hole, until it’s kept that way through the guide pin. The lower guides will release, and the guide pin is removed, completing the double knot.
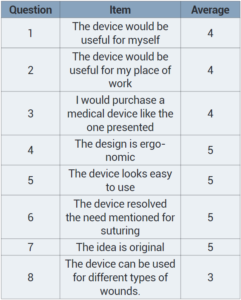
“After the prototype was assembled and design functions checked, the final step required a survey,” the team wrote. “The study contained questions about the medical device presented via prototype and they were asked to elaborate on their answers regarding their opinion as health professionals.”
The 3D printed prototype device was presented to Dr. Stephanie Gómez Najéra, Dr. Pamela Villareal Valverde, and Dr. Tatiana Piedra Chacón. The numbers listed in the survey results are the average between these three Costa Rican physicians, and the scale, based on the Likert scale, goes from 1-5, with 1 being strongly disagree and 5 being strongly agree.
“The comments reference that the usefulness depends on the context of where it would be applied, for example a jail or emergency room,” the researchers wrote of the doctors’ opinions on their device.
“One main drawback is that the device may not be suitable for all types of wounds. Other concerns raised by the physicians were related to the price and size of the device.”
Based on observations from the survey, the researchers modified the final prototype to “improve its ergonomic factor” by adding a holder at the top of the shell for more stability and easier manipulation.
Next steps include standardizing parts of the prototype so that some pieces can be purchased in the market, and optimizing the mechanisms, like the servomotor, sensors, and motors, that generate the device’s movements.
Discuss this story and other 3D printing topics at 3DPrintBoard.com or share your thoughts in the Facebook comments below.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
Ohio Ordnance Works Partners With Velo3D on Metal 3D Printing for Defense Initiative
Metal 3D printing firm Velo3D announced that it has partnered with Ohio Ordnance Works (OOW), a Class III arms manufacturer. The company makes the REAPR machine gun and SAW, BAR,...
Broad Spectrum Additive at the US Air Force
If we look at how Air Forces around the world use additive manufacturing, a lot of the attention is focused on future possibilities in hypersonics and next-generation aircraft. Beyond the...
EOS Releases Medical Device Master File for M290 3D Printer
Laser powder bed fusion (LPBF) firm EOS has released a Medical Device Master File that will make it easier for customers to achieve regulatory success. The file will essentially be...
Push Button Metal 3D Printing for $50,000?
In 2021 we wrote an article about “Push Button Metal, the low cost metal 3D printing evolution we are not talking about.” In it, we talked about One Click Metal,...