Testing Chemical Modifications and In-Vitro Biological Properties in FDM 3D Printing
In the recently published ‘Fused Deposition Modeling 3D Printing: Test Platforms for Evaluating Post-Fabrication Chemical Modifications and In-Vitro Biological Properties,’ international authors explore expanded uses for FDM 3D printing, creating polyvalent test plates (poly-lactic acid cylinders, 14 mm in diameter and 3 mm in height) for investigation of the biological properties of specific materials.
The authors remind us that 3D printing in the medical realm is most often centered around the fabrication of 3D printed models created from data like CT scans or MRIs. 3D printing has also been exponentially accentuated with the addition of accompanying technologies like robotics; however, for marketing more complex devices like implants, manufacturers must be able to pass numerous evaluations, meeting standards and regulations on multiple levels. Issues like microbial biofilm formation must be avoided, for example, leading to inflammatory responses and more in patients.
The researchers explain that through chemical modification negative types of growth can be inhibited.
“Interestingly, both growth inhibition and enhancement can be of considerable use to the researcher. A key point in such a modification is the presence of reactive ester groups in many of the polymers used in 3D printing, thus polylactic acid (PLA) and polyethylene terephthalate (PET) are based on polymers with ester backbones, whereas poly(methyl methacrylate) (PMMA) has ester side chain functions,” said the authors.
Ultimately, the goal of the research team was to find a connection between the study parameters and chemically modified PLA-based 3D printed polyvalent test plate platforms, or PVTPs. Samples were assessed using the following:
- Fourier transform infrared (FTIR) spectroscopy
- Contact angle measurements
- Scanning electron microscopy (SEM)
- Surface roughness measurement
- Positron annihilation lifetime spectrometry (PALS)
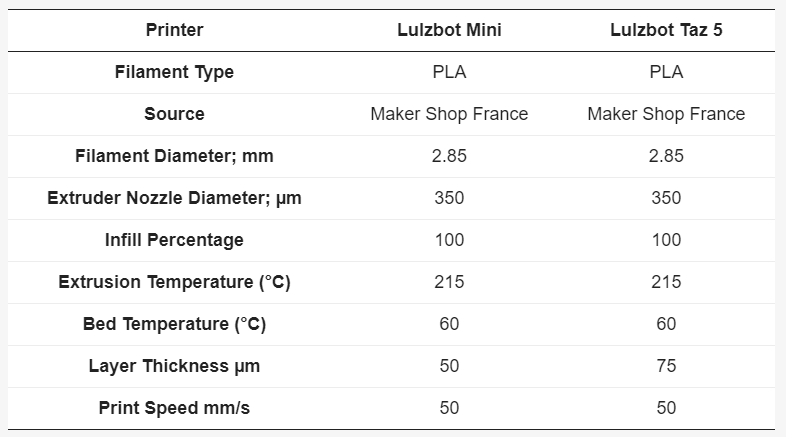
The researchers used ColorFabb PLA Naturel filament of 2.85 mm, along with both a Lulzbot Taz 5 3D printer (50 batches printed) and a Lulzbot Mini 3D printer (25 batches printed).
As with all FDM printing, initially adhesion happens to the print bed surface then to the previously printed layers, hence the importance of using the correct bed temperature for each source of filament.
PLA filaments were 3D printed at 215 °C through a 0.4 mm stainless steel nozzle, using a layer height of 50 µm or 75 µm and infill of 100 percent.

Top left STL (standard tessellation language) file format view of the polyvalent test plate (PVTP), top right printed PVTP showing size, bottom left 25 PVTP print batch on Lulzbot Mini, and bottom right 50 PVTP print batch on Lulzbot Taz 5.
Surface scanning was dependent on applied filters, evaluation areas, and more, with included testing of micron, submicron, and nano-roughness of surfaces. Positron annihilation spectroscopy (PALS) was responsible for measuring supramolecular structure of polymeric-based implants
“In our paper, we demonstrated the importance of surface characterization and cytocompatibility investigations of 3D printed PLA- PVTPs and chemically modified PLA-based PVTPs. The amines as side chains can favorably alter the surface properties, wettability, and biofilm formation of PLA PVTP. Low biofilm formation ability and favorable cytocompatibility profiles were also presented in the case of prolonged exposure. There are connections between the type and the other properties of PLA PVTPs. Overall, the properties of 3D printing materials can be dramatically enhanced by the modification of base polymers,” concluded the researchers.
“Based on these in-vitro tests (MTT and biofilm formation tests), it can be concluded that more than one assay should be used to determine cytotoxicity, so as not to over or underestimate the cytocompatibility of 3D printed PVTPs. However, cytotoxicity data alone are not necessarily predictive of in-vivo issues, but alongside other experiments (contact angle, PALS, and SEM results) the in-vivo compatibility data may be estimated.”
FDM 3D printing has been used in the creation of many medical models and devices, from oncology to infectious diseases. This type of 3D printing also has the potential to be a major player in the future of pharmaceutical production. What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com.
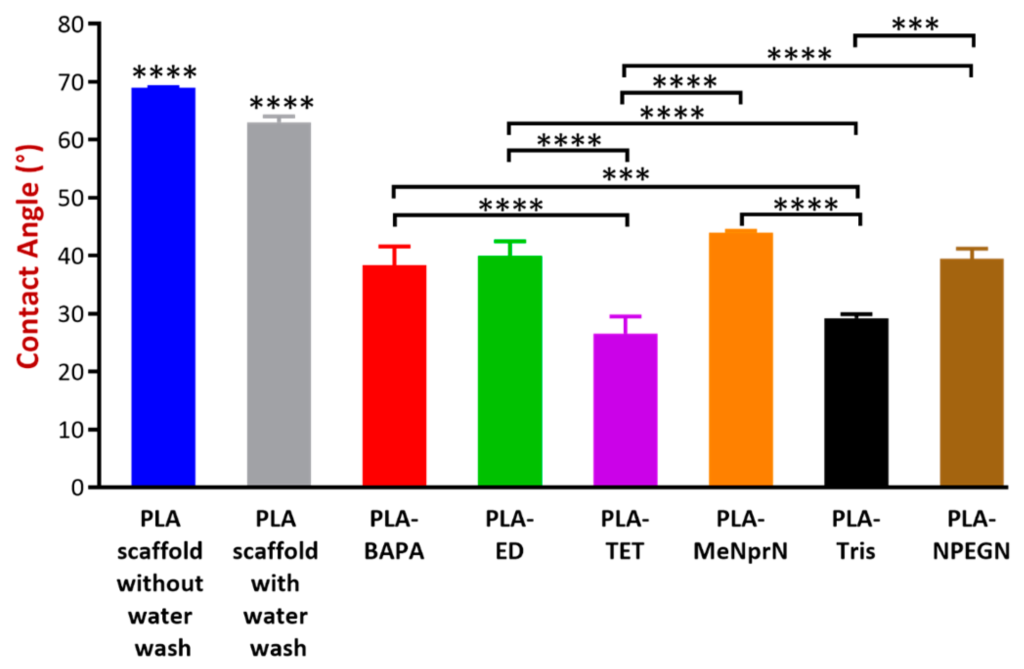
Contact angle values for the various PLA and chemically modified PVTPs. Data are expressed as means ± SD. Experiments were performed in triplicate, n = 3. Contact angle (°) values can be sorted in a decreasing order: PLA PVTP without water wash (69 ± 0.1°) = PLA PVTP with water wash (69 ± 0.1°) > PLA-MeNprN (45 ± 1.3°) > PLA-ED (41 ± 0.3°) > PLA-NPEGN (40 ± 1.1°) > PLA-BAPA (39 ± 0.5°) > PLA-Tris (30 ± 1.2°) > PLA-TET (26.5 ± 3.0°). The PLA PVTP with and without water wash have statistically significant differences in the contact angle values in comparison with the modified PLA based samples. PLA-TET and PLA-Tris modifications have statistically significantly lower results in comparison with the other modified PLA based samples. Error bars represent SD; *, **, and *** indicate statistically significant differences at p < 0.05, p < 0.01, and p < 0.001, respectively. In general, surfaces with contact angles of less than 90° are considered hemato-compatible.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
3D Printing Financials: 3D Systems Returns to Growth in Q1 2026
3D Systems (NYSE: DDD) reported one of its strongest quarters in recent years, showing signs that the company may finally be moving past the tough slowdown that has weighed on...
3D People Case Study Details Development of 3D Printed POV Camera Rig
A POV, or Point of View, camera rig, is a wearable support system that helps filmographers capture first-person footage, making the images more immersive. Some good examples of movies shot...
3D Printing News Briefs, May 2, 2026: Soft Robots, Agricultural Waste, & More
In this weekend’s 3D Printing News Briefs, we’ll start off with a multi-laser metal powder bed fusion 3D printer and post-processing news. We’ll end with research into soft robotics and...
Industrial Applications on Display at RAPID 2026: CERATIZIT & 3D Systems
Applications are where it’s at in the additive manufacturing (AM) industry. At the recent RAPID+TCT in Boston, I met with a few companies to learn about some of their very...