Chinese Startup Meditool Gets Investment From Evonik
 Last year Evonik opened a new production line for specialty PA [polyamide] powders at the Marl Chemical Park in Germany, increasing the company’s annual capacities by 50%. It also released a research hub for resource efficiency topics in Singapore, delving into topics such as additive manufacturing and functional surfaces. Now, the German company is extending its corporate venture capital activities into China with a single-digit million-Euro amount investment in 3D printing startup Meditool.
Last year Evonik opened a new production line for specialty PA [polyamide] powders at the Marl Chemical Park in Germany, increasing the company’s annual capacities by 50%. It also released a research hub for resource efficiency topics in Singapore, delving into topics such as additive manufacturing and functional surfaces. Now, the German company is extending its corporate venture capital activities into China with a single-digit million-Euro amount investment in 3D printing startup Meditool.
Headquartered at the Shanghai Industrial Technology Institute, Meditool makes implants for neuro and spine surgery and will support Evonik’s network and extend their presence in the Eastern subregion of Asia. Evonik is one of the world leaders in specialty chemicals, and investments like this also strengthen their Smart Materials growth engine with an innovative application for high-performance polymers.
China spends roughly 5% of their GDP on healthcare, well below the 10% world average, yet, the Chinese market is the world’s second-largest for medical implants with expected annual growth rates of over 10 percent. Meaning there is a growth opportunity for medical device companies, both local and foreign. But the Chinese government’s effort to control healthcare costs and foster the local medical device industry suggests possible market share gains for local brands, such as Meditool.
The medical device industry for the Asian giant was valued at $18.8 billion in 2016 and is projected to grow modestly through the remaining portion of this year when the value is expected to reach $24 billion. In the next few years, the country expects a growth rate of over 20 percent and projects the market to be valued at 600 billion yuan ($84.2 billion) in 2019.
“China is a key growth market for Evonik and one of the main drivers of innovation worldwide,” said Claas Klasen, Evonik’s President for the Northern Asia region. “It not only fosters leap-frog technology, it has the population and growing middle-class to drive fast-paced demand.”
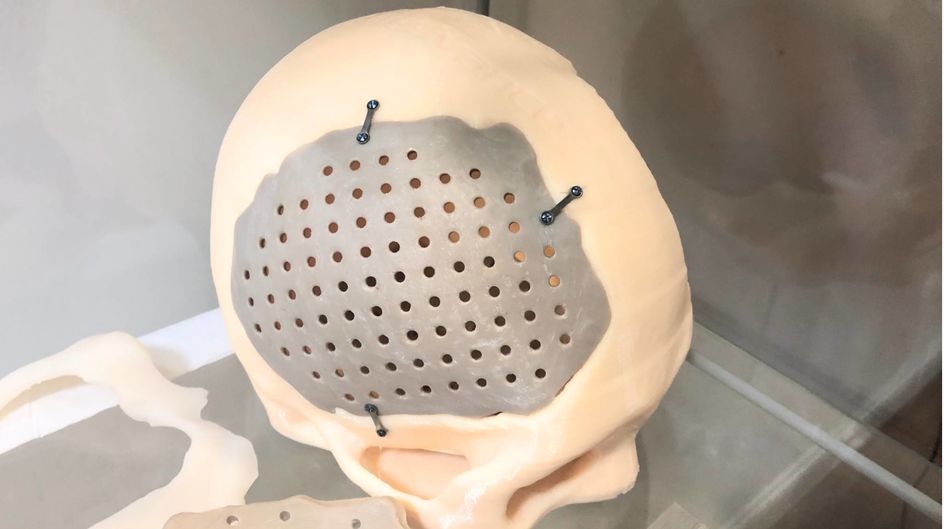
Meditool’s 3D printed neuro and spine surgery implants enable faster patient recovery, fewer post-operation checks for patients and less surgical risk for doctors. Plus, the company has developed its own hardware and software systems, which can read and process images directly from commonly used magnetic resonance imaging (MRI) or computed tomography scan (CT) devices. Once the readily printable 3D model is generated by the software, it sent to the printer. Meditool uses a high-performance polymer supplied by Evonik to 3D print the implants in medical grade Polyetheretherketone (PEEK), also marketed by the company under the name VESTAKEEP.
For patients and doctors, 3D printed PEEK implants are revolutionary compared with metal, the current conventional solution for the orthopedics implant market. The technology allows customization so that, for example, a plate fits perfectly in the patient’s skull. This reduces the likelihood that further operations will be required to adjust for size, shape or positioning of the implant. Other benefits include biocompatibility of the material and the possibility to carry out CT and MRI examinations after the surgery.
“Meditool is one of the pioneers in developing 3D printed PEEK medical implants and Evonik has been our trusted partner in materials supply. The venture investment will be an extra boost to our endeavor to bring innovative solutions to patients and surgeons in China and the rest of the world,” suggested Ken Jin, co-founder and chief technology officer of Meditool.
In 2016 Evonik began researching biodegradable composite materials as an alternative to metal implants. The research done at the company’s Medical Devices Project House in Birmingham, Alabama, was part of the company initiative into 3D printing biocompatible implants, which are an optimal alternative since they can be gradually absorbed by the body as the bone heals, eliminating the need for future surgeries.
“Meditool’s technology pays directly into our strategy of expanding in high-tech applications for our additive manufacturing materials,” explained Thomas Grosse-Puppendahl, head of Evonik’s innovation growth field in Additive Manufacturing. “Medical applications are of particular interest and our high-performance polymers have already been proven as a reliable implant material in other applications such as dental.”
Meditool has established a number of invention and utility model patents since its establishment, various partnerships with hospitals or medical related units in China, and even entered into a partnership with Canadian  raw material companies. From surgical models to 3D printing laboratories for in-hospital engineering design services, the start-up is providing an integrated medical 3D printing solution for healthcare providers.
raw material companies. From surgical models to 3D printing laboratories for in-hospital engineering design services, the start-up is providing an integrated medical 3D printing solution for healthcare providers.
Evonik’s venture capital arm has already invested in two funds in China and with Meditool, it now has its first direct investment. Co-investors in Meditool include ZN Ventures, Morningside Ventures, and Puhua Capital. Evonik VC plays a strategic role in Evonik’s goal to become a best-in-class specialty chemicals company by supporting a strong digital transformation. To this end, Evonik launched its second venture capital fund with €150 million at the beginning of 2019, more than doubling the amount under management to €250 million.
“This is our first direct investment in China and our first direct investment after initiating our second venture capital fund this year. Meditool is a good example of how venture capital is helping Evonik secure access to disruptive technologies,” claimed Bernhard Mohr, head of Evonik VC.
The investment in Meditool will help the company focus on areas that have a close fit to Evonik’s growth engines, areas which the company has identified as promising fields for future growth and development, as well as an excellent new proposal to a growing economy and medical implant market.
[Images: Evonik and Meditool]Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
Print Services
Upload your 3D Models and get them printed quickly and efficiently.
You May Also Like
AMPulse Asia: Creality IPO Headlines APAC 3D Printing Market Roundup
Asia’s additive manufacturing sector spent the back half of May moving capital and capacity, not just demos. Chinese desktop and consumer printer makers pushed onto public markets, metal powder producers...
Blue Origin’s New Glenn Explosion Comes During Major Manufacturing Push
Blue Origin‘s orbital New Glenn rocket exploded during a hot-fire test at Launch Complex 36 in Cape Canaveral on May 29, setting back the company’s launch ambitions at a time...
Aibuild Says New FETS Simulation Tool Is 10,000x Faster for AM
Aibuild has launched FETS for Additive Manufacturing, a Finite Element Thermomechanical Simulation tool that lets you simulate stress, distortion, thermal effects, and thermomechanical effects. The solution has been optimized for...
AI CAD Tools for 3D Printing: An Overview
There is a bevy of AI-to-CAD tools coming out. Some are finding users; some are raising millions in funding. Many new ones are coming out all the time, so we...