When it comes to 3D bioprinting with stem cells the obvious endgame is the ability to produce complex living tissue, such as replacement organs or biological matter. The potential future applications would completely change the world in numerous ways that are difficult to even predict. Transplant organs grown from a patient’s own DNA would be only one of the many uses of complex 3D bioprinted tissue; animal testing could be rendered completely unnecessary, as would the slaughter of animals as a food source. And while we are probably decades away from being able to produce even the simplest of living tissues, researchers and scientists are inching ever closer.
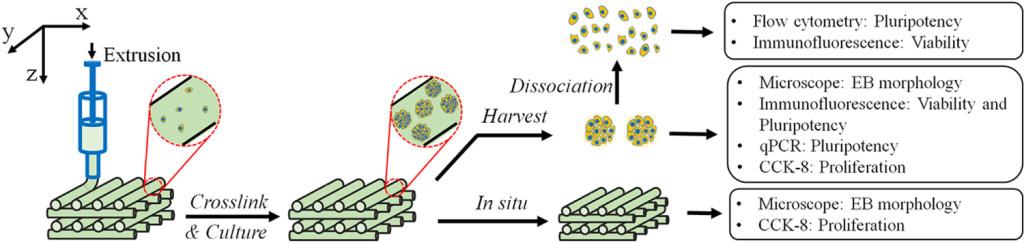
 A multinational group of researchers based at Tsinghua University in Beijing, China, and at Drexel University in Philadelphia have developed a bioprinting process capable of growing viable embryonic stem cell structures. The researchers used extrusion-based 3D printing technology to produce grid-like 3D structures that uniquely encouraged the growth of an embryoid body. These cell structures produced total cell viability and demonstrated rapid self-renewal for up to 7 days while exhibiting the ability to differentiate, or divide, into several different cell types.
A multinational group of researchers based at Tsinghua University in Beijing, China, and at Drexel University in Philadelphia have developed a bioprinting process capable of growing viable embryonic stem cell structures. The researchers used extrusion-based 3D printing technology to produce grid-like 3D structures that uniquely encouraged the growth of an embryoid body. These cell structures produced total cell viability and demonstrated rapid self-renewal for up to 7 days while exhibiting the ability to differentiate, or divide, into several different cell types.
Essentially, the test cell structures showed the potential of dividing and organizing themselves into complex, living tissue. While the tissue is typically only viable for a week, the fact that they were able to create stable groups of living cells for that length of time is remarkable. Other existing methods of growing stem cells were incapable of producing the types of cellular structures that the researchers were trying to grow, only the 3D printed method produced their desired results. A paper, titled ‘Three-dimensional bioprinting of embryonic stem cells directs highly uniform embryoid body formation,’ detailing the results of their 3D bioprinting process was just published in the medical journal Biofabrication. The paper’s authors included Liliang Ouyang, Rui Yao, Shuangshuang Mao, Xi Chen, Jie Na, and Wei Sun.
“It was really exciting to see that we could grow embryoid body in such a controlled manner. The grown embryoid body is uniform and homogenous, and serves as a much better starting point for further tissue growth. Two other common methods of printing these cells are either two-dimensional (in a petri dish) or via the ‘suspension’ method (where a ‘stalagmite’ of cells is built up by material being dropped via gravity.). However, these don’t show the same cell uniformity and homogenous proliferation. I think that we’ve produced a 3D microenvironment which is much more like that found in vivo for growing embryoid body, which explains the higher levels of cell proliferation,” explained the paper’s lead author, Wei Sun.
Bioprinting with stem cells, which are capable of turning into every cell type in the body, could potentially lead to the ability to build complex tissue structures and possibly even micro-organs. The team of researchers are also hopeful that their technique will be capable of producing an embryoid body quickly at high volumes. These quickly grown embryonic structures would allow other researchers the ability to perform tissue regeneration experiments or test the effects of drugs and chemicals on living tissue.
“Our next step is to find out more about how we can vary the size of the embryoid body by changing the printing and structural parameters, and how varying the embryoid body size leads to ‘manufacture’ of different cell types,” said another of the paper’s authors, Rui Yao.
The team is hoping to continue their research and expand the scope of their stem cell production process. First they want to learn how to alter the size and shape of the stem cells with simple adjustments to their bioprinting method. The next step would be studying exactly how those size changes would affect the cell’s development and differentiation, and if the grid pattern would produce the same results with the different sized cells. You can read the entire study online for free here.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
US Army Corps of Engineers Taps Lincoln Electric & Eaton for Largest 3D Printed US Civil Works Part
The Soo Locks sit on the US-Canadian border, enabling maritime travel between Lake Superior and Lake Huron, from which ships can reach the rest of the Great Lakes. Crafts carrying...
Construction 3D Printing CEO Reflects on Being Female in Construction
Natalie Wadley, CEO of ChangeMaker3D, could hear the words of her daughter sitting next to her resounding in her head. “Mum, MUM, you’ve won!” Wadley had just won the prestigious...
Blue Laser-powered M600 3D Printer Launched by Meltio
Founded in 2019 as a joint venture between Additec and Sicnova, metal 3D printer OEM Meltio develops and manufactures high-performance and easy-to-use metal 3D printing solutions that use its patented wire-laser metal...
3D Printed Storage Tanks Cut Material Costs by 25%
In a previous article, “Concrete Dreams: Let’s Print Money, Not Houses,” we discussed how the spotlight on 3D printing homes might be misplaced. Bollards, pedestrian bridges, and concrete tanks could...