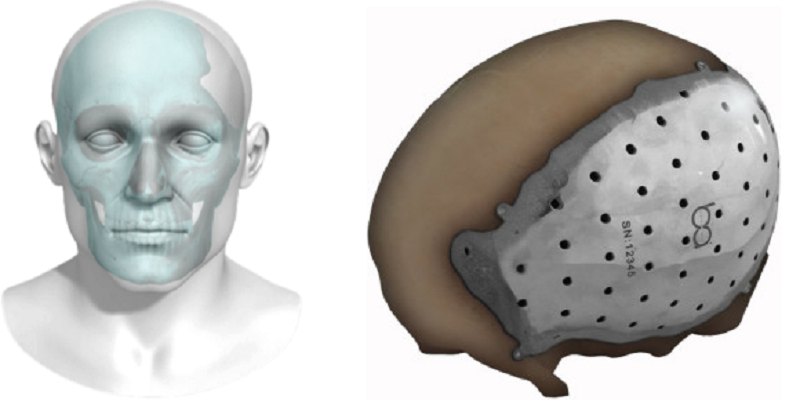
Today, BioArchitects announced that their 3D printed, patient-specific titanium cranial/craniofacial implant has been granted 510(k) clearance by the FDA. This is big news because it allows the company to begin marketing the device, the first of its kind in the United States.
“We are extremely proud to contribute to what we consider another major advance in the trend toward personalized medicine,” said Mark Ulrich, CEO of BioArchitects USA. “We believe that this is yet another step toward what will ultimately become the new standard of care.”
The devices are printed using electron beam melting (EBM) technology, patented by metal additive manufacturing corporation Arcam AB. The process, which – as its name suggests – uses electron beams to melt and fuse metal powders at high temperature and high speed, resulting in metal products that are lightweight, strong and incredibly precise – properties that have made the technology very popular in the manufacture of orthopedic devices and implants.
“BioArchitects is a prime example of how innovative organizations are using EBM technology to advance biomedical surgeries that truly effect people’s lives,” said Magnus René, CEO of the Arcam Group. “Arcam has been a strategic supplier to the orthopedic market for over a decade and tens of thousands of implants are made yearly from our EBM systems. It is clear that both BioArchitects and Arcam are advancing patient care with new technologies that will make a significant difference in the world of medicine for years to come.”
Discuss these new medical devices in the 3D Printed Titanium Implants forum over at 3DPB.com.