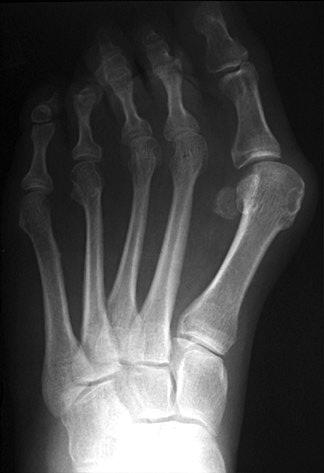
Hallux Valgus deformity, or as it’s more commonly known – a bunion – can be a debilitating foot ailment. It affects 23% of people age 18-65 each year, and it’s both painful and inconvenient to sufferers. To repair it, doctors have in the past had surgical options like fusing the first metatarsal or fusing the metatarsal-cuneiform joint as their most effective treatments. But patients who undergo those procedures face long recovery periods and a list of unpleasant complications.
Now a company called MedShape is using 3D printing and orthopedic device technology which uses what the company calls “proprietary shape memory technology” to give surgeons biomaterial tools which can transform and adapt inside the body.
MedShape, Inc. has now received U.S. Food and Drug Administration clearance for what they call their FastForward Bone Tether Plate, a device which uses 3D printing in a medical grade titanium alloy – Ti-6AL-4V – to fabricate human-implantable devices with complex and customizable geometries.
The plate is the key to what the company calls the FastForward Bunion Correction System. They say it preserves and protects native bone anatomy, and the system will debut this month at the American College of Foot and Ankle Surgeons (ACFAS) Annual Scientific Conference in Phoenix, Ariz.
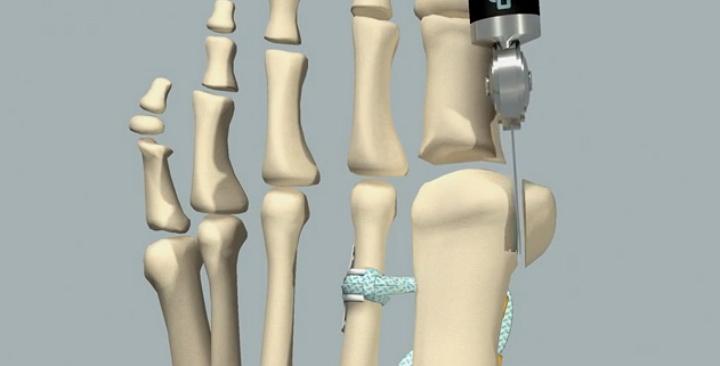 They say it’s a breakthrough in bunion correction which allows suture tape to be securely and safely wrapped around the second metatarsal. The technique eliminates the need to drill through the bone, and thanks to 3D printing technology, the Bone Tether Plate features unique design elements including a novel overall geometry which securely holds the plate in place.
They say it’s a breakthrough in bunion correction which allows suture tape to be securely and safely wrapped around the second metatarsal. The technique eliminates the need to drill through the bone, and thanks to 3D printing technology, the Bone Tether Plate features unique design elements including a novel overall geometry which securely holds the plate in place.
Dr. Ken Gall, the chief technology officer of MedShape and a professor of Materials Science and Engineering at the Georgia Institute of Technology, says the device and the FDA approval is key to the company’s efforts to develop and commercialize medical devices in new ways.
“We are in the early stages of tracking clinical outcomes with this new surgical approach and are excited to extend the 3D printing material platform towards the development of other implants,” says Gall.
MedShape, Inc. is a privately held medical device company focused on creating a portfolio of surgical solutions. The company says that the methods use patented new materials to improve treatments for sports medicine, joint fusion, and musculo-skeletal trauma products.
Have you heard of interesting medical advances which include 3D printing technology? Let us know in the 3D Printing and Aerospace Metals to Repair Foot forum thread on 3DPB.com.
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
3D Printing Financials: Fathom Struggles in Financial Quicksand During Critical Transition
Facing a year of key transitions and financial pressures, Fathom (Nasdaq: FTHM) has filed its annual report for 2023 with the U.S. Securities and Exchange Commission (SEC). The document outlines...
Latest Earnings Overview for Australian 3D Printing Firms Titomic and AML3D
Australian 3D printing manufacturing firms Titomic (ASX: TTT) and AML3D (ASX: AL3) reported their financial results for the period from July to December 2023, marking the first half of their...
3D Printing Webinar and Event Roundup: April 7, 2024
Webinars and events in the 3D printing industry are picking back up this week! Sea-Air-Space is coming to Maryland, and SAE International is sponsoring a 3D Systems webinar about 3D...
3D Printing Financials: Unpacking Farsoon and BLT’s 2023 Performance
In the Chinese 3D printing industry, two companies, Farsoon (SHA: 688433) and Bright Laser Technologies, or BLT (SHA: 688333), have recently unveiled their full-year earnings for 2023. Farsoon reported increases...