The Food and Drug Administration has a responsibility of protecting the public’s health, by assuring the safety of drugs, medical devices, and food supplies. Often times people forget that their other responsibility is to  stimulate innovation in order to advance the health of our citizens. These two initiatives often get in the way of each other, as innovation can’t occur if overregulation is present. Therefore the FDA has to take part in a balancing act, assuring safety while not stamping out innovation.
stimulate innovation in order to advance the health of our citizens. These two initiatives often get in the way of each other, as innovation can’t occur if overregulation is present. Therefore the FDA has to take part in a balancing act, assuring safety while not stamping out innovation.
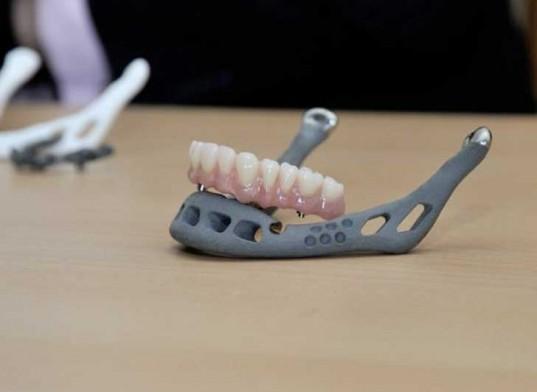
In just the last year alone, we have seen a tremendous amount of innovation within the field of medicine, thanks in part to the rapidly expanding 3D printing market. 3D printers are being used to create both internal and external medical devices, saving lives, and bettering the lives of literally thousands of people each year. At the same time though, the FDA has to assure the public that the medical community will not abuse this technology, putting our health at risk.
A notice dated May 14th, and written by Leslie Kux, the Assistant Commissioner for Policy for the FDA, has announced a public workshop to discuss the technical considerations of 3-D Printing, which will take place on October 8 and 9, 2014, from 8 a.m. to 5 p.m. The premise for the discussion, titled, “Additive Manufacturing of Medical Devices: An Interactive Discussion on the Technical Considerations of 3-D Printing”, is to gather comments from additive manufacturing companies, and medical device manufacturers on the problems, and solutions to those problems, that 3D printing will bring to the table. The workshop will be held at the FDA’s White Oak Campus, 10903 New Hampshire Ave., Bldg. 31 Conference Center, the Great Room (Rm. 1503), Silver Spring, MD. For those who wish to comment on the discussion at hand, but can’t make it to the campus, they will be able to submit comments electronically at regulations.gov.
The goals of this workshop, that the FDA hopes to achieve are as follows:
Understand
It is very important for those within the industry as well as those working for the FDA to understand the technology as well as the problems and challenges that lie ahead. 3D printing is a relatively new technology, thus there are numerous issues which may not have even been considered yet.
Create Awareness
As a group, the participants of this workshop will be able to collaborate on solutions to any problems which may be present, and develop the best practices to ensure the safety of 3D printed medical devices.
Coordinate/Promote
Innovation is one of the FDA’s main goals. Through this workshop, they seek to establish processes which will ensure performance and reliability of 3D printed devices. They also seek to spark collaboration between those within the industry, on developing educational materials, standards, and guidance.
It will likely be a couple of years before any regulations emerge, stemming from this planned public discussion. Creating regulations which are fair, balanced, and most importantly protect the public, can be a long process. Hopefully the FDA ultimately finds a middle ground between overregulation, and turning a blind eye. Share your opinions on what the FDA’s job should be in regulating 3D printing at the FDA forum thread on 3DPB.com
Subscribe to Our Email Newsletter
Stay up-to-date on all the latest news from the 3D printing industry and receive information and offers from third party vendors.
You May Also Like
IperionX Inks 10-Year Deal with Wisconsin Manufacturer for 80 Metric Tons of Titanium Per Year
IperionX, the Charlotte-based supplier of sustainable titanium powders used for additive manufacturing (AM) and metal injection molding (MIM), has signed a ten-year deal with United Stars, a group of industrial...
Gastronology Launches Industrial Production of 3D Printed Food for Dysphagia Patients
Food 3D printing has, in many ways, been an additive manufacturing (AM) segment looking for the right business case. While some applications are beautiful and others may or may not...
Lockheed Martin Leads $3M Investment in Q5D’s Electronics 3D Printing System
Q5D, an original equipment manufacturer (OEM) of robotic arm, hybrid additive manufacturing (AM) systems used for wire harness production, has closed a $3 million investment round. The investment arm of...
3D Printing News Briefs, April 6, 2024: Depowdering, Cybertruck Door Handles, & More
In today’s 3D Printing News Briefs, ioTech’s digital manufacturing CLAD technology is opening up opportunities for microelectronics and additive manufacturing. Hexagon and Raytheon Technologies commercially released the Simufact Additive Process...